Dominant Role of Clonal Deletion in Achieving Immune Tolerance After Unrelated Cord Blood Transplantation.
1BMT and Cell Therapy, Children Hospital of Pittsburgh UPMC, Pittsburgh, PA
2Immunology, University of Pittsburgh, Pittsburgh, PA
Meeting: 2017 American Transplant Congress
Abstract number: 198
Keywords: Bone marrow transplantation, Graft-versus-host-disease, T cell clones, Tolerance
Session Information
Session Name: Concurrent Session: Tolerance: Clinical Studies
Session Type: Concurrent Session
Date: Sunday, April 30, 2017
Session Time: 4:30pm-6:00pm
 Presentation Time: 5:18pm-5:30pm
Presentation Time: 5:18pm-5:30pm
Location: E353C
The mechanism of immune tolerance after successful hematopoietic stem cell transplantation is not fully understood. Both central (clonal deletion) and peripheral (anergy, Treg, Tr1) mechanisms are possible. In this study, we tested these mechanisms after unrelated cord blood transplant (UCBT) in the GvH direction. Methods 7 patients (age 1m to 9y) with non-malignant diseases were transplanted and enrolled on a reduced intensity conditioning (RIC) trial (NCT01852370). 5 patients were off immunosuppression therapy (IST) with no GVHD had fully immunocompetent while 2 had either limited or recently resolved GvHD and were still on IST when studied. Donor T cell chimerism was a median 97%. Purified T cells responses to host APCs were measured by mixed lymphocyte reaction (MLR) and cytotoxic lymphocyte (CTL) reaction, along with cytokine secretion profiling. To track the fate of host-reactive T cell clones, TCRß repertoire was analyzed using ImmunoSEQ. To identify the key mechanisms of tolerance, we depleted Tregs with Ontak treatment prior to MLR and CTL reactions. Involvement of Tr1 or anergy was examined either by blocking IL10R or by adding low dose IL2, respectively. Results and Discussion: In patients with clinical tolerance, there was no significant proliferative or cytotoxic T cell response towards host APC while T cells responded vigorously to 3rd party APC (Fig1). Similarly, cytokine profiles revealed recipient-specific non-responsiveness (Fig1A), fulfilling a critical tenet of tolerance in three independent assays. In addition, tracking TCRß clonal profiles revealed the disappearance of recipient-specific T cell clones that were identified from the cord blood graft itself pre-UCBT (Fig1C). There was no indication of Treg or Tr1 involvement in sustaining tolerance. In summary, the rapid acquisition of immune tolerance in the GvH direction post-UCBT on our RIC clinical trial is characterized by hypo-reactivity towards host APC. The proliferation, cytotoxicity, cytokine secretion and ImmunoSEQ data suggest that host-specific clonal deletion is a significant mechanism of tolerance.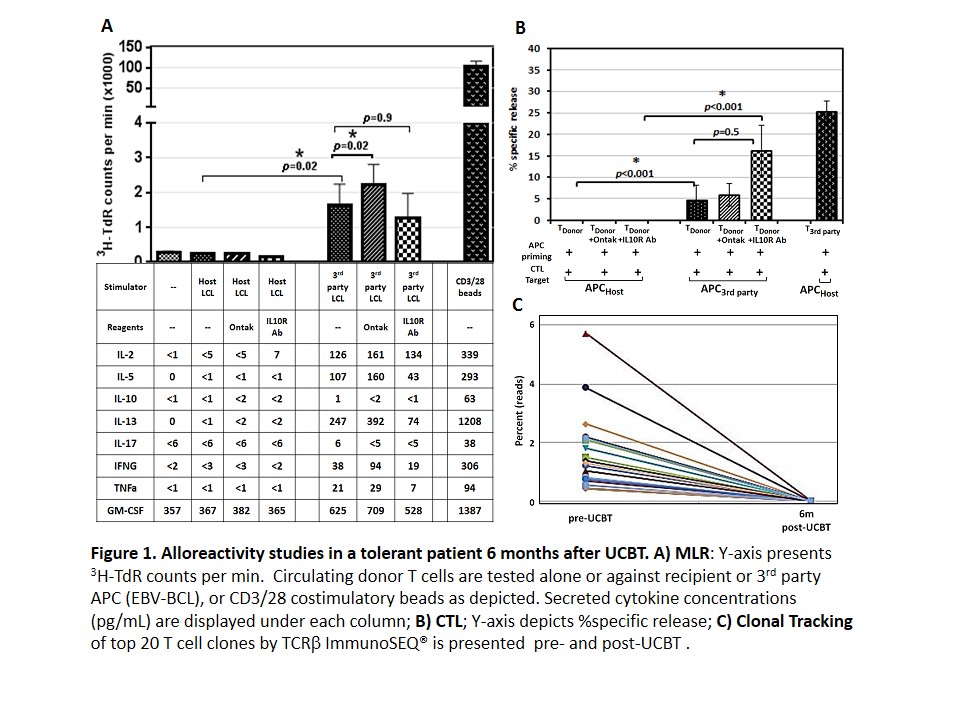
CITATION INFORMATION: Szabolcs P, Chen X, Hill M. Dominant Role of Clonal Deletion in Achieving Immune Tolerance After Unrelated Cord Blood Transplantation. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Szabolcs P, Chen X, Hill M. Dominant Role of Clonal Deletion in Achieving Immune Tolerance After Unrelated Cord Blood Transplantation. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/dominant-role-of-clonal-deletion-in-achieving-immune-tolerance-after-unrelated-cord-blood-transplantation/. Accessed May 14, 2026.« Back to 2017 American Transplant Congress
