Oxidative Stress Induces Cytoprotective Alternative Splicing in Carcinoembryonic Antigen Related Cell Adhesion Molecule 1 in Mouse Liver Ischemia-Reperfusion Injury
Surgery, Dumont-UCLA Transplant Center, UCLA, Los Angeles, CA
Meeting: 2020 American Transplant Congress
Abstract number: 538
Keywords: Adhesion molecules, Gene expression, Ischemia, Oxidant stress
Session Information
Session Name: Cellular Therapies, Tissue Engineering / Regenerative Medicine
Session Type: Oral Abstract Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:45pm
 Presentation Time: 4:15pm-4:27pm
Presentation Time: 4:15pm-4:27pm
Location: Virtual
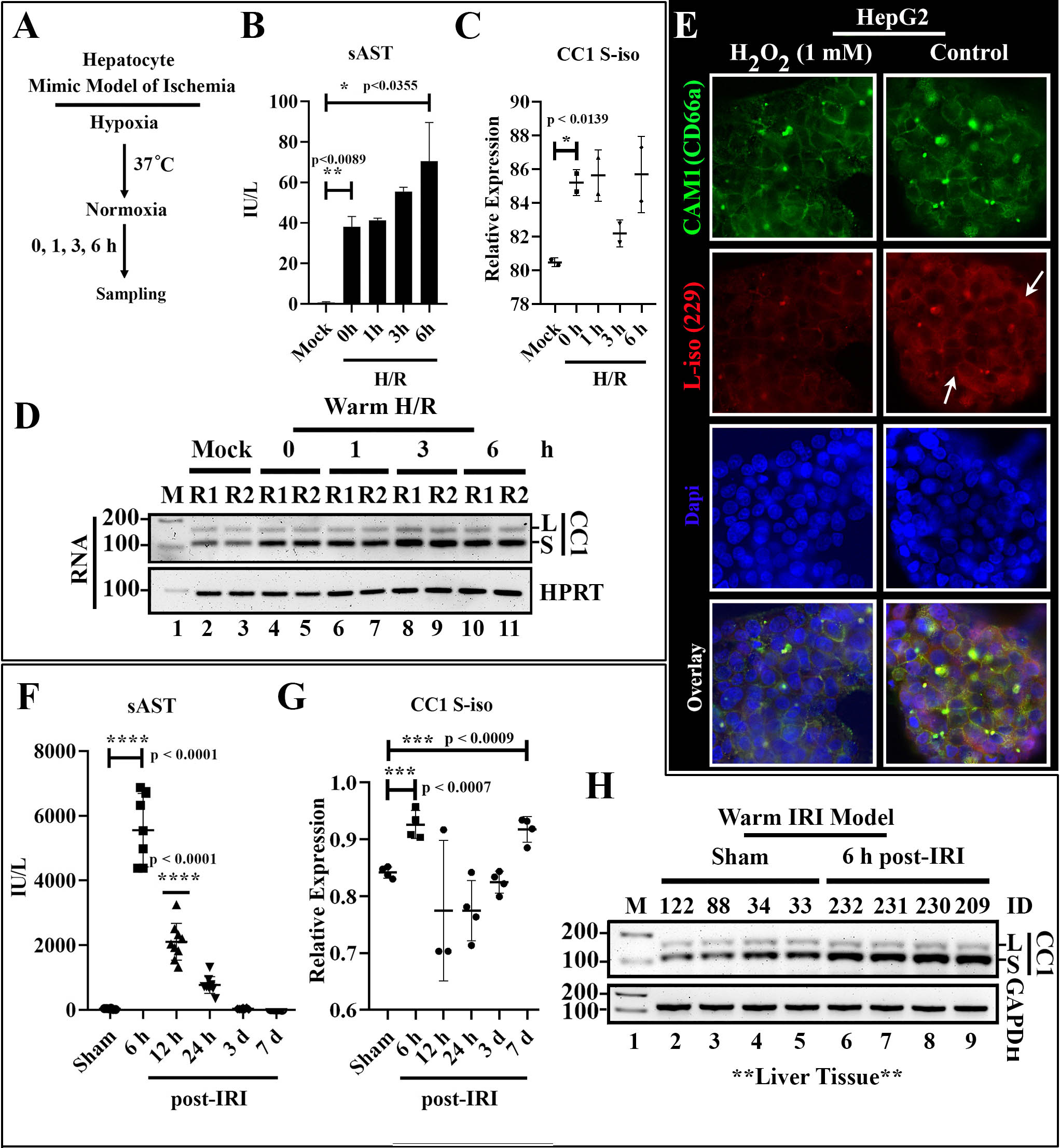
*Purpose: We have recently shown that carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) serves as a checkpoint regulator in liver transplant (LT) ischemia-reperfusion injury (IRI); a potential denominator of donor hepatic tissue quality; and a novel target for therapeutic modulation in LT recipients. However, as CEACAM1 undergoes extensive alternative splicing (AS) to generate functionally distinct S-iso and L-iso, it is currently unclear which of these two cytoplasmic isoforms are involved at the interface of IRI-LT immune injury and metabolic homeostasis.
*Methods: Primary mouse hepatocytes and HepG2 cells, derived from a human liver cancer cell line, were cultured using an in vitro inflammation-mimic model of hepatic warm IRI. Cells were preconditioned with a cytokine mix that included TNF-α, IFN-γ, IL-1β and endotoxin LPS exposure, followed by hypoxia/reoxygenation (H/R) or H202 treatment (1 mM). Splicing isoforms of CEACAM1 were monitored by RT-PCR, immunoblotting, and immunofluorescence. Longitudinal in vivo analyses of CEACAM1 AS was monitored in a murine model of liver warm ischemia (60min) followed by reperfusion (6h, 12h, 24h, 3d, and 7d).
*Results: In the in vitro arm, primary mouse hepatocytes exposed to H/R readily induced CEACAM1 AS with prolonged exposure to IRI conditions (at 6h), and S-iso production was accompanied by the highest sAST levels (mean±SEM 69.75 ± 13.50, p<0.0355). Similarly, H202-stressed HepG2 cells showed preferential CEACAM1 AS towards S-iso protein expression phenotype. In the in vivo arm, mouse hepatic tissues from an innate immune-driven inflammation model of warm IRI showed the kinetic production of S-iso, which peaked at 6 h post-reperfusion (n=4; mean±SEM 0.08407 ± 0.01333, p<0.007) and reemerged at day 7 (n=4; mean±SEM 0.07566 ± 0.01233, p<0.0009), demonstrating that AS was triggered by hepatic IR-stress and that S-iso was required for the resolution of tissue inflammation. In contrast, L-iso was maximally expressed between 12h and 24h, post-reperfusion. Since immunofluorescence studies have revealed comparable hepatic neutrophil infiltration patterns (Ly6G staining), it appears that the expression of L-iso did not result from CEACAM1 AS.
*Conclusions: This study is the first to identify the oxidative stress and hepatic tissue injury as catalytic drivers of CEACAM1 alternative splicing; while the production of its S-isoform might mediate cytoprotection and/or control donor transplant IR-stress resistance.
To cite this abstract in AMA style:
Dery KJ, Hirao H, Kadono K, Kageyama S, Kojima H, Busuttil RW, Kupiec-Weglinski JW. Oxidative Stress Induces Cytoprotective Alternative Splicing in Carcinoembryonic Antigen Related Cell Adhesion Molecule 1 in Mouse Liver Ischemia-Reperfusion Injury [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/oxidative-stress-induces-cytoprotective-alternative-splicing-in-carcinoembryonic-antigen-related-cell-adhesion-molecule-1-in-mouse-liver-ischemia-reperfusion-injury/. Accessed May 14, 2026.« Back to 2020 American Transplant Congress