HTFPI.hCD47 and Adhesion Inhibition in GTKO.hCD46 Pig Lung Xenograft Injury
1Center for Transplantation Sciences, Massachusetts General Hospital, Charlestown, MA, 2Revivicor, Inc, Blacksburg, VA
Meeting: 2021 American Transplant Congress
Abstract number: 593
Keywords: Adhesion molecules, Gene expression, Graft survival, Lung transplantation
Topic: Basic Science » Xenotransplantation
Session Information
Session Name: Xenotransplantation
Session Type: Poster Abstract
Session Date & Time: None. Available on demand.
Location: Virtual
*Purpose: We evaluate whether genetic expression of human tissue factor pathway inhibitor (TFPI, extrinsic coagulation pathway inhibitor) and human CD47 (“don’t eat me” immunoregulator), alone or with combined PSGL-1 (selectin) and Mac-1 (integrin) (“adhesion inhibition”), influences GalTKO.hCD46 porcine lung injury during perfusion with human fresh blood.
*Methods: Expression of TFPI and CD47 was confirmed by qRT-PCR and immunochemistry; TFPI function on pAECs was assessed using a thrombin generation assay. In a well-established paired ex vivo lung perfusion model, GTKO.hCD46.hTFPI.hCD47 transgenic pig lungs (n=5) were compared to reference GTKO.hCD46 lungs (n=5). All lungs were treated with a thromboxane synthase inhibitor (1-BIA), anti-histamine (Famotidine), and anti-GPIb integrin-blocking Fab. In both genotypes, one lung of each pair was additionally treated with adhesion inhibitors (n=4 hTFPI.hCD47, n=3 reference).
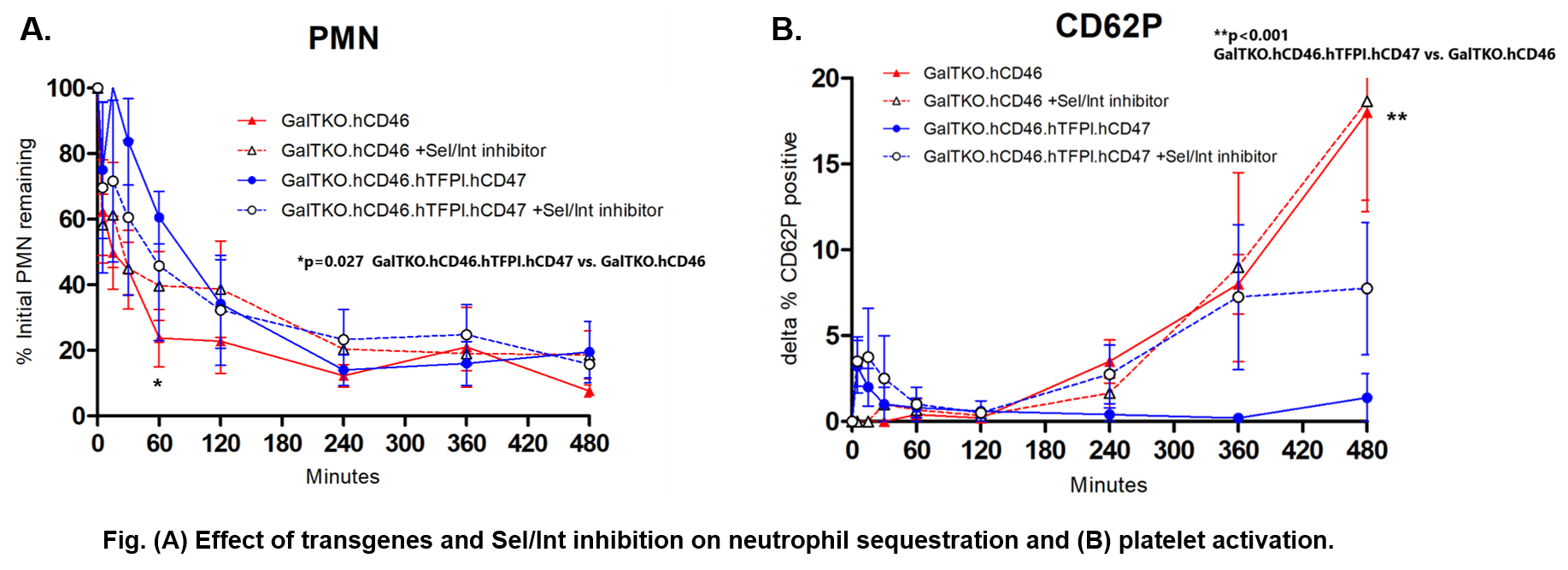
*Results: In vitro, GTKO.CD46.TFPI.CD47 pAEC triggered less thrombin formation compared to GTKO (normalized AUC: 93 +/- 2 vs. 100 +/- 0.4, p<0.005). Ex vivo, all except for two GalTKO.hCD46 lungs (183, 440 min) survived until elective termination at 8 hours. Additional adhesion inhibition moderately lowered PVRs in lungs expressing hTFPI.hCD47 during the first 2 hours. Neutrophil sequestration was significantly delayed during the first hour (P<0.05) in association with hTFPI.hCD47 expression, but not with adhesion inhibition (Fig.A). Lungs expressing hTFPI.hCD47 significantly attenuated terminal platelet activation measured by CD62P expression compared to GTKO.CD46 (P<0.001) (Fig.B). Additional adhesion inhibitors didn't further suppress platelet activation in GalTKO.hCD46 or GalTKO.hCD46.hTFPI.hCD47-expressing lungs.
*Conclusions: Expression of hTFPI.hCD47 showed protective effects (prolonged survival, delayed initial neutrophil sequestration and attenuated platelet activation) under these study conditions. While additional adhesion inhibitor reduced PVR elevation associated with hTFPI.hCD47 expression, it didn’t significantly attenuate neutrophil or platelet sequestration by the lung. We conclude that non-canonical adhesive mechanisms mediate the adhesion of human formed blood elements to pig endothelium that occurs even in the context of multiple genetic modifications and drug treatments designed to attenuate lung xenograft injury by these pathways.
To cite this abstract in AMA style:
Miura S, Burdorf L, Habibabady Z, Dandro A, Connolly M, Pratts S, Phelps C, Eyestone W, Ayares D, Azimzadeh A, III RPierson. HTFPI.hCD47 and Adhesion Inhibition in GTKO.hCD46 Pig Lung Xenograft Injury [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/htfpi-hcd47-and-adhesion-inhibition-in-gtko-hcd46-pig-lung-xenograft-injury/. Accessed May 22, 2026.« Back to 2021 American Transplant Congress