Extra-Renal Cardiac Injury Is Associated with Upregulation of NOD2 and Nlrp3 in the Heart After Renal IRI.
Medicine, Division of Nephrology and Hypertension, University of California San Diego, La Jolla, CA
Meeting: 2017 American Transplant Congress
Abstract number: 217
Keywords: Heart, Immunogenicity, Inflammation, Renal ischemia
Session Information
Session Name: Concurrent Session: Emerging Interventions in Ischemia Reperfusion Injury
Session Type: Concurrent Session
Date: Monday, May 1, 2017
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:30pm-3:42pm
Presentation Time: 3:30pm-3:42pm
Location: E351
Background: Recent studies show renal ischemia reperfusion injury (IRI) is associated with systemic release of danger associated molecular patterns (DAMPs). DMAPs induce significant extra-renal organ injury by activating pattern recognition receptors (PRRs) on immune cells and parenchymal cells in several extra-renal organs, including the heart. Binding of IRI-associated DAMPs to PRRs stimulates proinflammatory cytokines and cell death signaling in cardiac myocytes and fibroblasts. In turn, activated cardiac cells release DAMPs, leading to a cascade of ongoing immune damage to the heart following renal IRI.
Purpose: Our study sought to assess the injurious effects of renal IRI-induced inflammation on cardiac tissue.
Methods: Hearts were isolated from WT mice (C57BL/6) subjected to nonlethal renal IRI (15min IRI). Quantitative PCR and WB assessed changes in expression of innate immune receptors and markers of apoptosis and necrosis in cardiac tissue of injured vs. uninjured mice. FACS assessed leukocyte infiltrates in hearts, over time.
Results: Testing for changes in PRR expression, we saw marked upregulation of NOD2 and Nlrp3 in hearts after renal IRI. The upregulation was sustained for 14 weeks after initial injury (Figure 1) and was associated with ongoing apoptosis of cardiac myocytes and fibroblasts, suggesting a prolonged effect on the heart. Cardiac tissue from mice subjected to renal IRI exhibited robust and sustained upregulation of NOD2 and Nlrp3, but not TLR2, TLR4, NOD1, ASC or caspase-1. Associated with PRR activation was ongoing infiltration of inflammatory cells (CD4, CD8 T cells, B cells, Macrophages and DCs) into the cardiac tissue up to 14 weeks after renal IRI.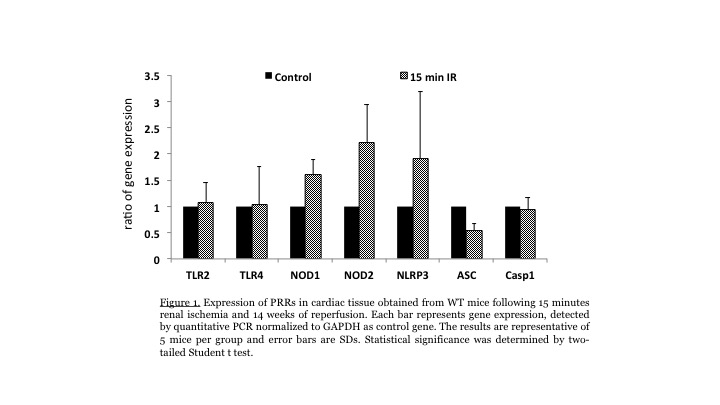 Conclusions: Our data suggest renal IRI triggers a prolonged inflammatory response in the heart. The inflammatory response is associated with upregulation of NLRs (NOD2 and Nlrp3) and NLR-mediated apoptosis, which is sustained over time. These data suggest that NLR activation might be an important target for amelioration of cardiac damage associated with renal IRI.
Conclusions: Our data suggest renal IRI triggers a prolonged inflammatory response in the heart. The inflammatory response is associated with upregulation of NLRs (NOD2 and Nlrp3) and NLR-mediated apoptosis, which is sustained over time. These data suggest that NLR activation might be an important target for amelioration of cardiac damage associated with renal IRI.
CITATION INFORMATION: Scheinok A, Kasimsetty S, Soo E, Nelson C, Shigeoka A, McKay D. Extra-Renal Cardiac Injury Is Associated with Upregulation of NOD2 and Nlrp3 in the Heart After Renal IRI. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Scheinok A, Kasimsetty S, Soo E, Nelson C, Shigeoka A, McKay D. Extra-Renal Cardiac Injury Is Associated with Upregulation of NOD2 and Nlrp3 in the Heart After Renal IRI. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/extra-renal-cardiac-injury-is-associated-with-upregulation-of-nod2-and-nlrp3-in-the-heart-after-renal-iri/. Accessed May 18, 2026.« Back to 2017 American Transplant Congress
