Development and Cell Surface Testing of a Novel Nanofiber Technology for Targeted Delivery of Therapeutic Regulatory T Cells
1Surgery -Transplant, Northwestern University, Chicago, IL, 2Simpson Querrey Institute, Northwestern University, Chicago, IL
Meeting: 2020 American Transplant Congress
Abstract number: D-293
Keywords: Bioengineering, Immunosuppression, Preclinical trails, Tolerance
Session Information
Session Name: Poster Session D: Cellular Therapies, Tissue Engineering / Regenerative Medicine
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
*Purpose: Regulatory CD4+CD25+FOXP3+ T cells (Tregs) have been shown to induce tolerance in transplantation models, and to be increased in their numbers in tolerant human solid organ transplant recipients. Therefore, a number of centers including ours have pursued the large-scale isolation and expansion of clinical (GMP) grade Tregs and are testing them in clinical organ transplant recipients. For Treg therapy to be most effective for tolerance induction, the infused cells need to migrate to the transplanted organ. Peptide amphiphile (PA) nanofibers are being used to coat individual cells for their protection, targeted delivery, and controlling biological functions. Targeted delivery of Tregs may have the desired effect of enhancing efficacy and reducing the number of cells required for therapeutic effect.
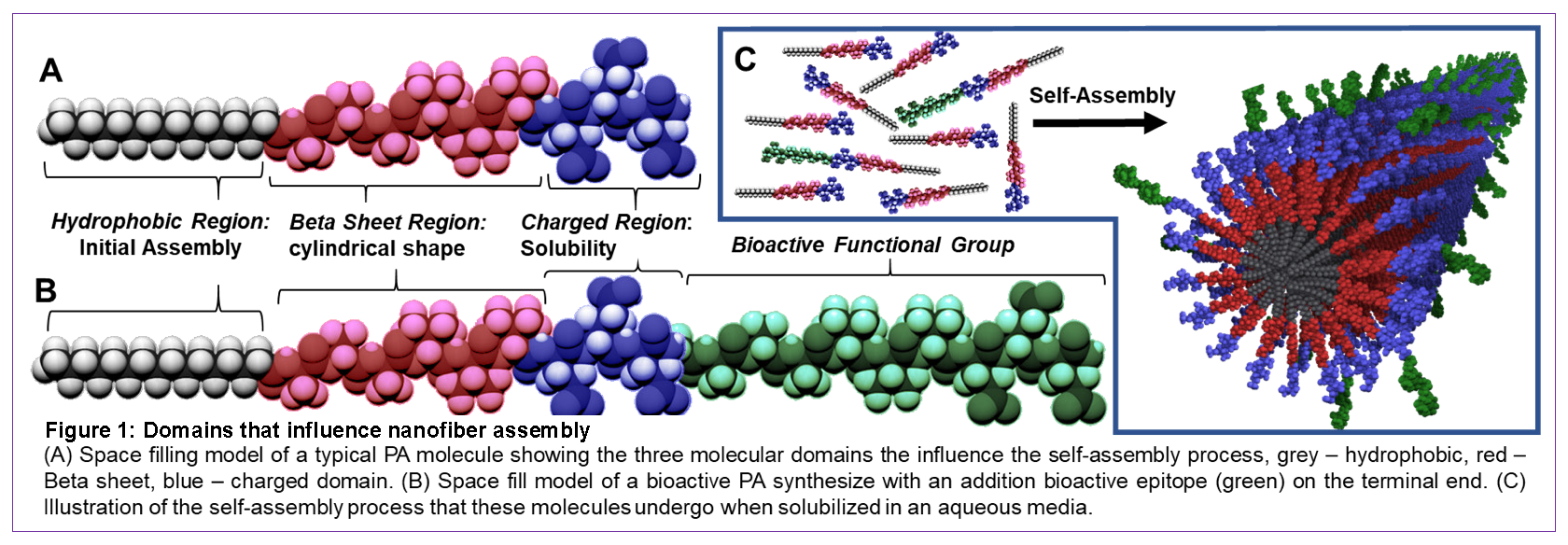
*Methods: We have developed a novel nanofiber technology consisting of self-assembling PA molecules of C16-VVVAAAEEE that can temporarily bind to cells and therapeutic agents. The PA (Figure 1A) were found to coat a wide range of cells efficiently, without interrupting their natural phenotype or functions. The use of targeting bioactive peptide sequence in the PA (Figure 1B) can also deliver the coated cells to specific tissue sites
*Results: In this study, we have bioengineered PA displaying a VCAM-1 binding epitope, VHPKQH (VCAM-b-PA) and scrambled QHPHVK control (VCAM-scr-PA). We then assessed their targeting abilities in vitro using human expanded Tregs. VCAM-1 is a major molecule involved in trans-endothelial migration of cells including Tregs to the inflamed/transplant site. VCAM-b-PA could coat Tregs at significantly lower concentrations than the PA or VCAM-scr-PA, and none of the PAs were toxic to the cells. Tregs coated with VCAM-b-PA and not with VCAM-scr-PA demonstrated specific binding to VCAM-1. Overnight culture of coated Tregs, however, resulted in the internalization of the VCAM-PAs into the cytosol, thus suggesting that the coating is transient but long enough for the cells to reach the target tissue. More importantly, Treg phenotype and suppressive function were maintained following coating with the nanofibers.
*Conclusions: These results indicated that this strategy has great potential for targeted delivery of Tregs to areas of interest such as inflamed tissues and transplanted organs where they can readily function as immune regulators.
To cite this abstract in AMA style:
Mathew JM, Lee S, Sambandam Y, Fraser LA, Clemons TD, Kolberg A, He J, Palmer LC, McClendon MT, Zhang ZJ, Leventhal JR, Stupp SI, Gallon L. Development and Cell Surface Testing of a Novel Nanofiber Technology for Targeted Delivery of Therapeutic Regulatory T Cells [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/development-and-cell-surface-testing-of-a-novel-nanofiber-technology-for-targeted-delivery-of-therapeutic-regulatory-t-cells/. Accessed May 30, 2026.« Back to 2020 American Transplant Congress