Versatile Strategy to Enhance Nanomedicine Delivery to Graft Endothelial Cells
1Yale University, new haven, CT, 2Perlmutter Cancer Center, New York University, Langone Medical Center, New York, NY, 3University of Cambridge, Cambridge, United Kingdom
Meeting: 2021 American Transplant Congress
Abstract number: 528
Keywords: Efficacy, Endothelial cells, Kidney, Machine preservation
Topic: Basic Science » Cellular Therapies, Tissue Engineering/Regenerative Medicine
Session Information
Session Name: Cellular Therapies, Tissue Engineering/Regenerative Medicine
Session Type: Poster Abstract
Session Date & Time: None. Available on demand.
Location: Virtual
*Purpose: Efficient delivery of therapeutics to graft endothelial cells (EC) can potentially render organs more resistant to injury, improving clinical outcomes. Specific targeting of polymeric nanoparticles (NPs) during ex vivo machine perfusion prior to transplant can allow both sustained delivery and increased local concentration of an encapsulated therapeutic. Despite ready access, effective retention by ECs lining graft vessels remains as a significant hurdle. Here we describe a new targeting approach based on oriented presentation of a targeting antibody using a linker called a “monobody” (Mb).
*Methods: Mbs are synthetic binding proteins with a unique cysteine engineered to enable site-specific conjugation to NPs through thiol chemistry. Specific Mbs can potently and selectively bind to the Fc region of a targeting antibody (Ab), forming Ab-Mb-NP conjugates. We used flow cytometry and quantitative fluorescence microscopy of dye-loaded NPs to measure efficiency of binding to ECs in cell culture and in ex-vivo perfusion systems of single vessels and of transplant-declined human kidneys.
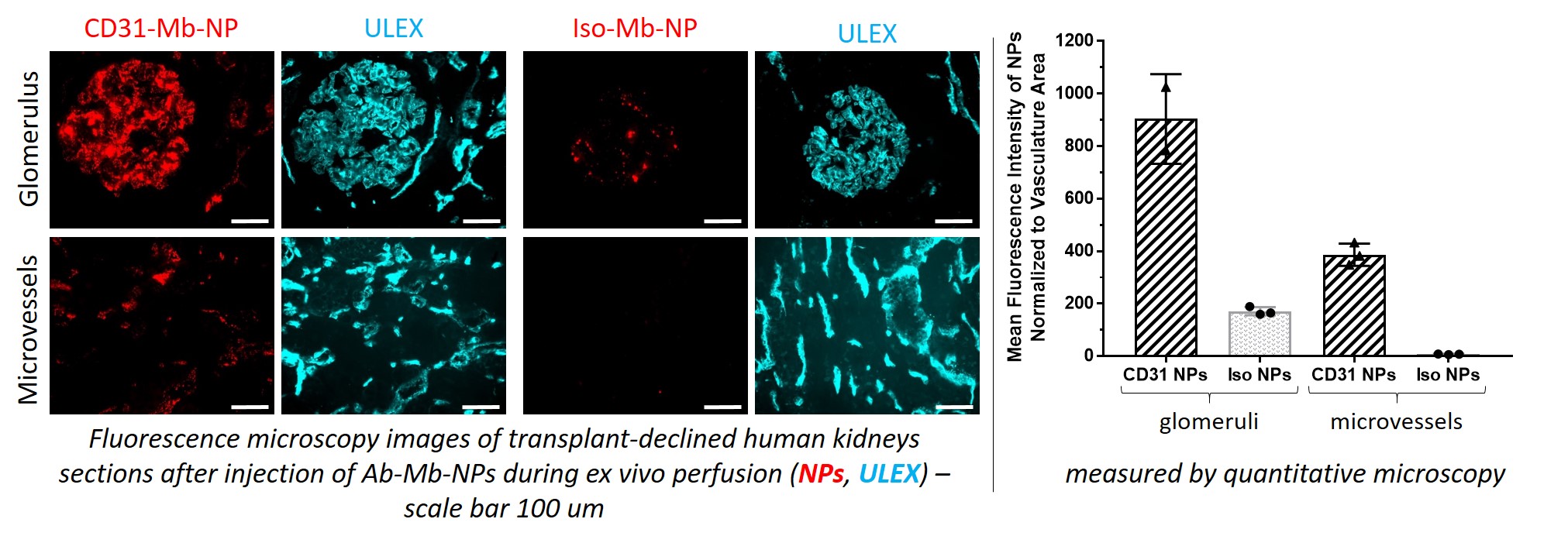
*Results: Ab-Mb-NPs showed up to a 1000-fold enhancement of binding in vitro under flow compared to EDC-NHS conjugated Ab-NPs used in our prior work. This is likely due to a higher number of Ab attached and a better control of Ab orientation leading to high retention of antibody function. In the first transplant-declined human kidneys enrolled in the study, the targeted Ab-Mb-NPs bound specifically to the endothelial cells covering ~40% of the microvessels and ~70% of the glomeruli vasculature area. In addition, the Mb approach is readily adaptable to different NP compositions or conjugation of different Abs of the same species/isotype without re-engineering as is necessary with EDC/NHS conjugation. For example, we easily changed the polymer used to formulate the NPs (poly(lactic acid)-poly(ethylene glycol) [PLA-PEG] or poly(amine-co-ester) [PACE]). We also easily changed the targeted molecule (CD31 or ICAM2) or the species of the targeted molecule (human or pig) using identical conditions for conjugation.
*Conclusions: Mb-coupled conjugation can both simplify and enhance the use of nanomaterials to target graft ECs, opening opportunities to efficiently deliver therapeutics prior to transplantation.
To cite this abstract in AMA style:
Albert C, Bracaglia LG, Koide A, DiRito J, Lysyy T, Edwards C, Langford JT, Haakinson D, Hosgood SA, Nicholson ML, Pober JS, Saltzman WM, Kiode S, Tietjen G. Versatile Strategy to Enhance Nanomedicine Delivery to Graft Endothelial Cells [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/versatile-strategy-to-enhance-nanomedicine-delivery-to-graft-endothelial-cells/. Accessed May 18, 2026.« Back to 2021 American Transplant Congress