Resolution of Hyperglycemia Following Islet Allograft Transplantation in Non-Immunosuppressed Mice
1Center for Transplantation Sciences, Massachusetts General Hospital, Boston, MA, 2Pathology, Massachusetts General Hospital, Boston, MA
Meeting: 2019 American Transplant Congress
Abstract number: 368
Keywords: B cells, Immunosuppression, Islets, Tolerance
Session Information
Session Name: Concurrent Session: Islet and Cell Transplantation
Session Type: Concurrent Session
Date: Monday, June 3, 2019
Session Time: 4:30pm-6:00pm
 Presentation Time: 5:06pm-5:18pm
Presentation Time: 5:06pm-5:18pm
Location: Room 309
*Purpose: Kidney allografts are naturally tolerogenic in mice. Certain mouse strain combinations develop tolerance of kidney allografts across full MHC incompatibilities without immunosuppression (e.g. DBA/2 to B6) while other combinations do not (e.g. B6 to DBA/2). This tolerance is initially dependent Foxp3+ cells that are concentrated in the graft in distinctive Treg-rich organized lymphoid structures (TOLS). Recipients of allograft kidneys develop systemic tolerance to tolerant-resistant heart allografts in pigs, non-human primates, and mice. The plan of this study is to identify the cellular and molecular mechanisms of kidney induced islet allograft tolerance (KI2AT). Using a co-transplantation model of kidney and islet in mice, we aim to test if spontaneous tolerance of mouse kidney graft is able to induce systematic tolerance to islet grafts in the recipient.
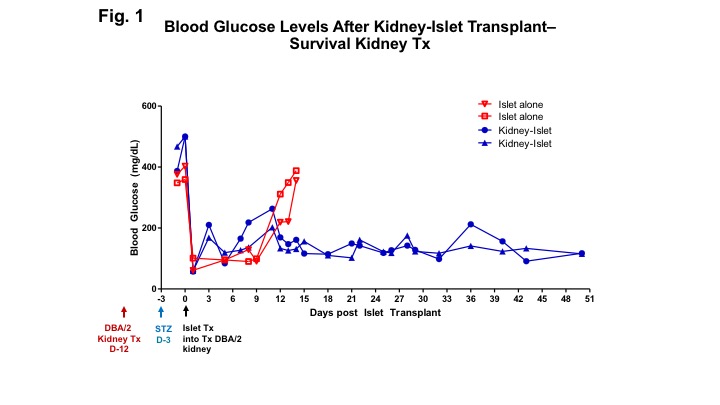
*Methods: We present preliminary data that show that spontaneously accepted DBA/2 kidney allografts in bilaterally nephrectomized recipients can confer tolerance to DBA/2 islets, transplanted under the kidney capsule of the renal allograft, resolving hyperglycemia in streptozotocin (STZ)-induced diabetic B6 mice.
*Results: We show that the spontaneous acceptance of kidney allografts confers systemic tolerance to islet allografts (Fig. 1). At day 50, the kidney allografts were removed and analyzed pathologically. H&E staining of the kidney/islet allografts show a massive aggregation of leukocytes at the kidney-islet junction (Fig. 2). Immunohistochemical staining showed that, while CD3+ and Foxp3+ cells are present within these aggregates, the majority of these cells are B cells (Fig. 3).
*Conclusions: The findings from this project provide a model that will lead to more therapeutic regimens that can be translated to the clinic, incorporating cellular and molecular components shown to be essential for the development of systemic tolerance resulting in the acceptance of islet allotranplants, independent of co-transplantation with kidney allografts.
To cite this abstract in AMA style:
Ge J, Lee K, Yang C, White R, Rosales I, Russell P, Rickert C, Markmann J, Madsen J, Colvin R, Alessandrini A. Resolution of Hyperglycemia Following Islet Allograft Transplantation in Non-Immunosuppressed Mice [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/resolution-of-hyperglycemia-following-islet-allograft-transplantation-in-non-immunosuppressed-mice/. Accessed May 22, 2026.« Back to 2019 American Transplant Congress