Preventing Cytomegalovirus Infection in Intermediate-High Risk Liver Transplant Recipients: Acyclovir Vs Valganciclovir
S. Dulanya, A. Anamisis, D. Tsapepas.
Department of Pharmacy, New York Presbyterian Hospital, New York City, NY.
Meeting: 2015 American Transplant Congress
Abstract number: C132
Keywords: Cytomeglovirus, Infection, Liver transplantation, Viral therapy
Session Information
Session Name: Poster Session C: Liver Retransplantation and Other Complications
Session Type: Poster Session
Date: Monday, May 4, 2015
Session Time: 5:30pm-6:30pm
 Presentation Time: 5:30pm-6:30pm
Presentation Time: 5:30pm-6:30pm
Location: Exhibit Hall E
Cytomegalovirus (CMV) is an opportunistic infection responsible for profound morbidity and mortality among solid organ transplant recipients. A donor and recipient's CMV IgG serostatus influences risk for development of CMV infection. Risk is considered “intermediate-high” for seropositive recipients of allografts from seropositive donors (D+/R+). The purpose of this study was to evaluate the effectiveness of two prophylactic strategies: acyclovir and valganciclovir among intermediate-high risk patients.
Single center, retrospective study of adult orthotopic liver transplant (OLT) recipients transplanted from Sept 2007–Mar 2013. All patients received ganciclovir 5mg/kg IV every 12hrs for 7 days post-OLT followed by two eras of prophylaxis – Era1 (from 9/07-12/10): acyclovir 800mg 3 times daily or Era2 (from 1/11-3/13): valganciclovir 900mg daily. The primary endpoint was the incidence of CMV viremia. Secondary endpoints included time to viremia and patient survival.
Of the 713 OLT performed during the study period, 157 were intermediate high-risk. Demographic variables were similar between groups and summarized in Table 1.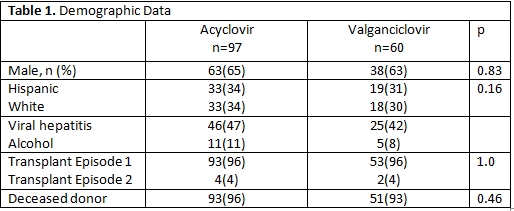 Viremia occurred in significantly more Era1 patients (30%) vs Era2 (8%), p=0.001. Mean time to viremia was significantly shorter in Era1(84 days) vs Era2(190 days),p=0.0006.
Viremia occurred in significantly more Era1 patients (30%) vs Era2 (8%), p=0.001. Mean time to viremia was significantly shorter in Era1(84 days) vs Era2(190 days),p=0.0006.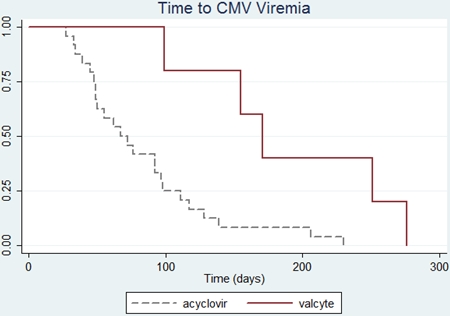 Era2 patients had higher rates of survival (95%),compared to Era1(71%),p<0.0001.
Era2 patients had higher rates of survival (95%),compared to Era1(71%),p<0.0001.
Use of acyclovir in the first era of prophylaxis showed higher rates of viremia with a faster onset and decreased patient survival. In contrast, valganciclovir was more effective in preventing viremia, prolonging its onset and enhancing patient survival. Valganciclovir should be continued as the main agent for CMV prophylaxis in intermediate-high risk patients.
To cite this abstract in AMA style:
Dulanya S, Anamisis A, Tsapepas D. Preventing Cytomegalovirus Infection in Intermediate-High Risk Liver Transplant Recipients: Acyclovir Vs Valganciclovir [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/preventing-cytomegalovirus-infection-in-intermediate-high-risk-liver-transplant-recipients-acyclovir-vs-valganciclovir/. Accessed May 17, 2026.« Back to 2015 American Transplant Congress
