Dependency on CD4 T Cells, Foreign Protein, and CD22 for Generation of Antibody Response to Non-Self Blood Group A-Antigen in a Mouse Model
Alberta Transplant Institute, Canadian National Transplant Research Program, University of Alberta, Edmonton, AB, Canada.
Meeting: 2018 American Transplant Congress
Abstract number: 178
Keywords: Antibodies, B cells, T cells, Tolerance
Session Information
Session Name: Concurrent Session: B-cell / Antibody
Session Type: Concurrent Session
Date: Monday, June 4, 2018
Session Time: 2:30pm-4:00pm
 Presentation Time: 2:54pm-3:06pm
Presentation Time: 2:54pm-3:06pm
Location: Room 615/616/617
Background: ABO-incompatible heart transplantation (ABOi-HTx) is safe during infancy and allows increased donor access. B cell tolerance develops to donor A/B-antigens (Ag) after ABOi HTx by unknown mechanisms. We developed transgenic mice (A-Tg) expressing human A-Ag on vascular endothelium and erythrocytes (RBC) to study anti-A antibody responses. CD22 participates in B cell tolerance and we found that human B cells express high-levels of CD22 and it decreases with age. Here we used a mouse model to study anti-A response in the context of syngeneic, allogeneic and xenogeneic stimulation, and the impact of CD22 expression.
Methods: Part I: Adult wild type (WT) C57BL/6, BALB/c, C3H/He, or CD22-deficient B6 (CD22KO) mice received injections of B6 or BALB A-Tg blood cells or human group-A (hu-A) RBC membranes (10% v/v), or A-incompatible heart allograft. Anti-A Ab was measured by hemagglutination; graft survival was assessed by palpation. Part II: a) To assess requirement of foreign protein to stimulate anti-A, hu-O RBC mixed with syngeneic A-Tg cells or allogeneic A-Tg blood were co-injected in WT mice; b) to assess T cell dependency, CD4+ T cells were depleted before hu-A RBC injection. Part III: To assess the role of CD22, A-Tg or hu-A-RBC, were injected into CD22KO mice with or without CD4+ T-cell depletion.
Results: Part I: Exposure to allogeneic A-Tg blood, heart graft, or xenogeneic hu-RBC induced anti-A production, whereas syngeneic A-Tg blood did not. Part II: a) mixture of syngeneic A-Tg/hu-O RBC did not induce anti-A; b) after CD4+ T-cell depletion, hu A-RBC failed to elicit anti-A. Part III: Hu A-RBC induced a very high anti-A in CD22KO mice compared to WT B6. In contrast to WT B6 mice, anti-A Ab was elicited in CD22KO mice following injection with A-Tg blood cells or hu A-RBC with CD4+ T cell depletion.
Conclusions: Our results show that in WT mice, anti-A production requires CD4+ T cells; consistent with a T-dependent anti-A response. Conversely, in CD22KO mice there was no requirement for CD4+ T cells; consistent with a T-independent anti-A response. 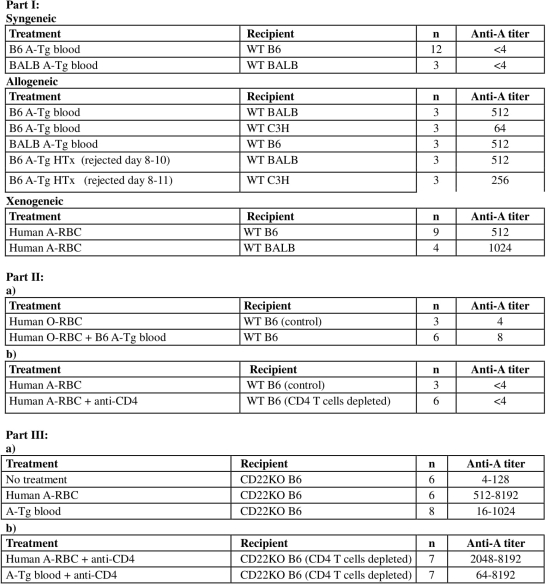
CITATION INFORMATION: Adam I., Motyka B., Tao K., West L. Dependency on CD4 T Cells, Foreign Protein, and CD22 for Generation of Antibody Response to Non-Self Blood Group A-Antigen in a Mouse Model Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Adam I, Motyka B, Tao K, West L. Dependency on CD4 T Cells, Foreign Protein, and CD22 for Generation of Antibody Response to Non-Self Blood Group A-Antigen in a Mouse Model [abstract]. https://atcmeetingabstracts.com/abstract/dependency-on-cd4-t-cells-foreign-protein-and-cd22-for-generation-of-antibody-response-to-non-self-blood-group-a-antigen-in-a-mouse-model/. Accessed May 14, 2026.« Back to 2018 American Transplant Congress
