A Clinically Relevant Protocol for Vascularized Composite Allotransplantation Using a Single Dose of AMD3100 for Stem Cell Mobilization.
1Surgery, Division of Plastic and Reconstructive Surgery, University of Colorado School of Medicine, Aurora, CO
2Clinical Research Division, Fred Hutchinson Cancer Research Center, Seattle, WA
3Medicine, University of Washington Medical Center, Seattle, WA
Meeting: 2017 American Transplant Congress
Abstract number: B299
Keywords: Engraftment, Graft survival, Stem cells, Tolerance
Session Information
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall D1
BACKGROUND: Vascularized Composite Allograft (VCA) transplantation is a clinical reality but application is limited by toxicities of chronic immunosuppression and rejection. Current clinical tolerance protocols limit use to living donor transplants. We sought to design a clinically relevant protocol applicable to cadaveric organs. We modified our existing non-myeloablative stem cell canine VCA transplant model to use AMD3100 for stem cell mobilization.
METHODS: 4 DLA-haploidentical related canine recipients [Group I] received conditioning with 350cGy TBI, AMD3100-mobilized donor stem cells and VCA transplantation with a short course of immunosuppression. 6 DLA-haploidentical related canine recipients [Group II] underwent identical conditioning plus an infusion of Bone Marrow (BM) harvested on the day of transplant. 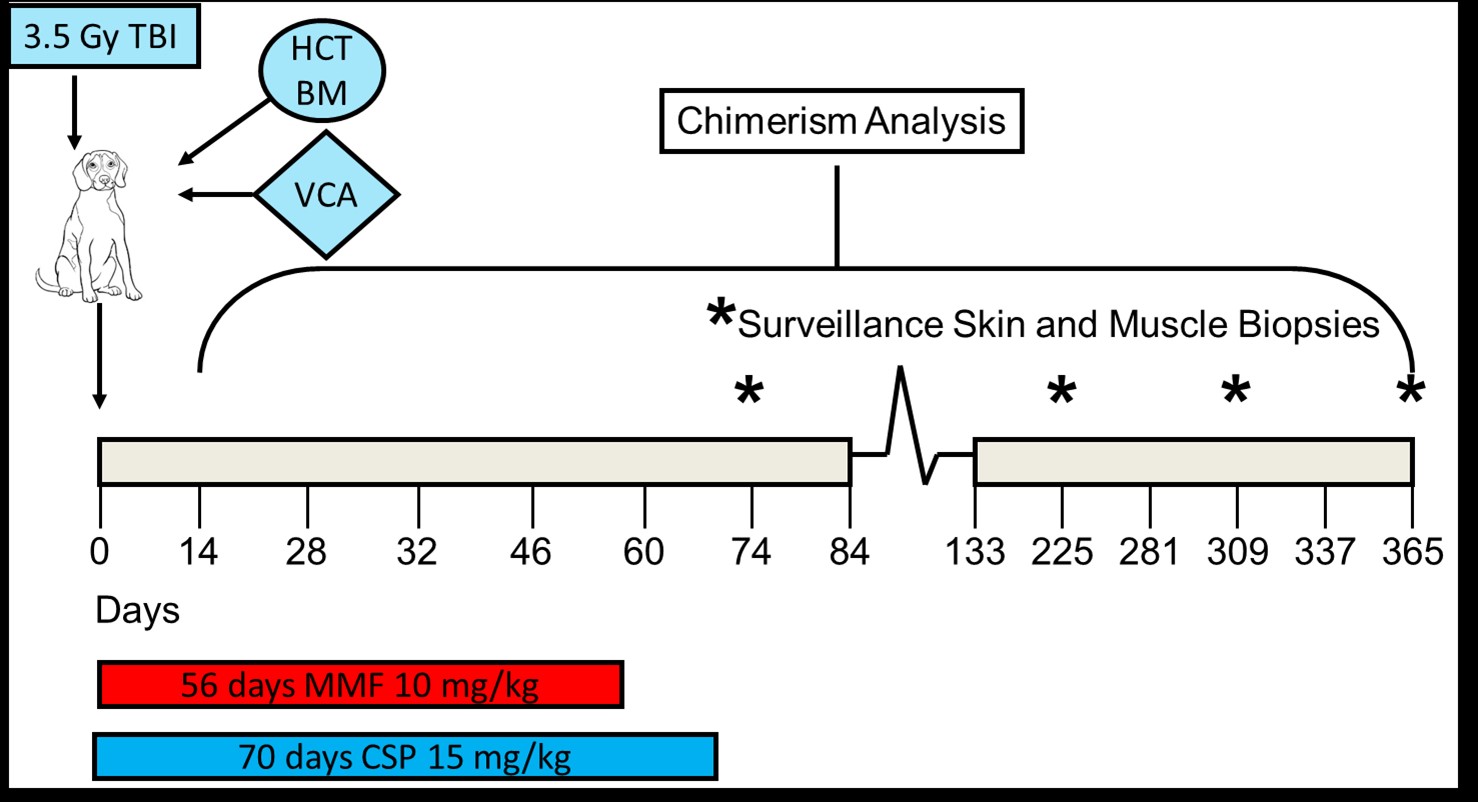 CD34+ hematopoietic progenitor cells were quantified via flow cytometry. Peripheral blood chimerism was evaluated by PCR techniques weekly. VCA graft survival was followed clinically and histologically.
CD34+ hematopoietic progenitor cells were quantified via flow cytometry. Peripheral blood chimerism was evaluated by PCR techniques weekly. VCA graft survival was followed clinically and histologically.
RESULTS: All dogs in the first group exhibited prolonged thrombocytopenia and 1 dog was euthanized secondary to this complication (POD 32). All 4 demonstrated initial stem cell engraftment. One dog had very poor initial engraftment and went on to reject the VCA on POD 146. Remaining 2 dogs remained tolerant to their VCA (POD 79; 101). The addition of BM eliminated the problems with prolonged thrombocytopenia. In Group II, 2 dogs were euthanized secondary to pneumonia (POD 12; 95) and 1 for liver dysfunction (POD 45). Currently 3 dogs are doing well with no evidence of GVHD or loss of the VCA transplant (POD 211; 92; 24).
CONCLUSION: This study demonstrates that a clinically relevant protocol using a single dose of AMD3100 and addition of a bone marrow aspirate combined with our non-myeloabative protocol can lead to tolerance the VCA across a significant genetic barrier.
CITATION INFORMATION: Swearingen B, Graves S, Storb R, Mathes D. A Clinically Relevant Protocol for Vascularized Composite Allotransplantation Using a Single Dose of AMD3100 for Stem Cell Mobilization. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Swearingen B, Graves S, Storb R, Mathes D. A Clinically Relevant Protocol for Vascularized Composite Allotransplantation Using a Single Dose of AMD3100 for Stem Cell Mobilization. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/a-clinically-relevant-protocol-for-vascularized-composite-allotransplantation-using-a-single-dose-of-amd3100-for-stem-cell-mobilization/. Accessed May 30, 2026.« Back to 2017 American Transplant Congress
