Use of Microbial Cell Free DNA Sequencing in Solid Organ Transplant Patients
S. Chang, P. Gaynor, A. Multani, O. E. Beaird, M. Carlson, S. Yang, O. Garner, J. Schaenman
University of California, Los Angeles, Los Angeles, CA
Meeting: 2021 American Transplant Congress
Abstract number: 725
Keywords: Genomics, Non-invasive diagnosis
Topic: Clinical Science » Infectious Disease » All Infections (Excluding Kidney & Viral Hepatitis)
Session Information
Session Name: All Infections (Excluding Kidney & Viral Hepatitis)
Session Type: Poster Abstract
Session Date & Time: None. Available on demand.
Location: Virtual
*Purpose: Microbial cell free DNA (cfDNA) sequencing has emerged as a new noninvasive tool for the diagnosis of infectious diseases in recent years, but its diagnostic utility remains in question. We sought to assess this by characterizing its usage in solid organ transplant (SOT) patients.
*Methods: We performed a retrospective chart review of all SOT patients who had microbial cfDNA sequencing done from 2016-2020.
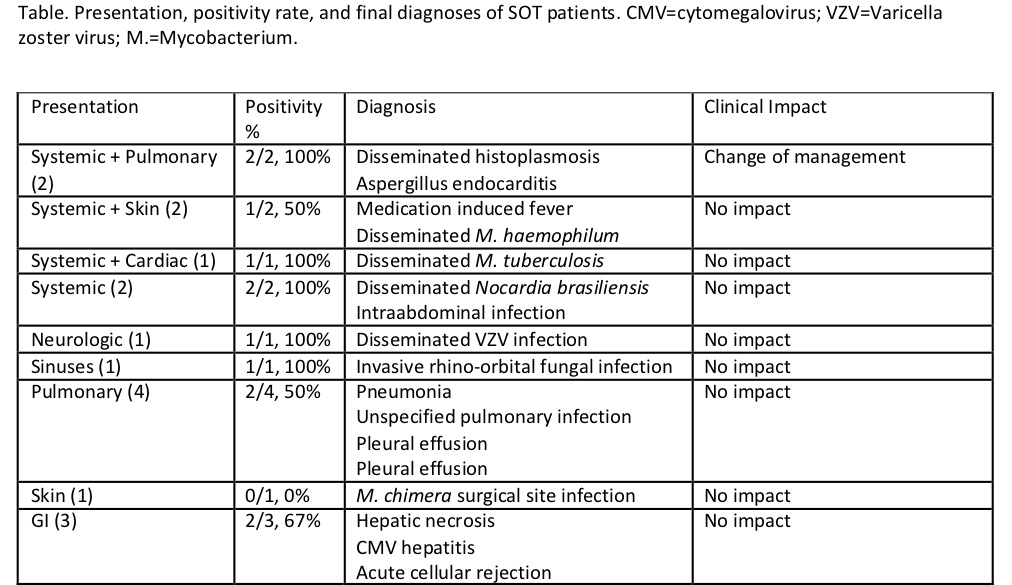
*Results: There were 17 adult SOT patients who underwent microbial cfDNA sequencing, including 6 kidney, 3 lung, 5 liver, 2 intestinal, and 1 heart transplants. Presentations that prompted testing included systemic, neurologic, sinuses, pulmonary, skin, gastrointestinal, and combinations of the above (see Table). Positivity rate for microbial cfDNA sequencing was 12/17 (70.6%), with a sensitivity of 66.7%. The highest positivity was found in patients who had systemic, neurologic, and sinus infections. Of the positive tests, 2 led to a change in either management or diagnosis (2/12, 16.7%). One of the two cases was a kidney transplant patient with disseminated histoplasmosis in which the cfDNA sequencing preceded positive cultures by 12 days. The second case was a liver transplant patient who was diagnosed with Aspergillus endocarditis based on the cfDNA sequencing result when conventional lab methods resulted negative. The remaining 10 cases with positive sequencing results did not lead to changes in management nor diagnosis due to either earlier results via conventional lab methods or lack of clinical correlation.
*Conclusions: In this solid organ transplant cohort, microbial cfDNA appeared to have highest positivity rate in select group of patients, and led to clinical changes in 16.7% of cases. Given the importance of early and accurate diagnosis in immunocompromised patients, cfDNA based diagnostics hold promise as a method for noninvasive testing. Future studies will explore the strengths and limitations of this testing approach in different clinical scenarios to better delineate appropriate testing algorithms.
To cite this abstract in AMA style:
Chang S, Gaynor P, Multani A, Beaird OE, Carlson M, Yang S, Garner O, Schaenman J. Use of Microbial Cell Free DNA Sequencing in Solid Organ Transplant Patients [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/use-of-microbial-cell-free-dna-sequencing-in-solid-organ-transplant-patients/. Accessed May 20, 2026.« Back to 2021 American Transplant Congress