Tissue Engineering Human Kidneys for Costimulation Blockade
1BREONICS, Inc., Watervliet, NY
2Surgery, University of Utrecht, Utrecht, Netherlands.
Meeting: 2015 American Transplant Congress
Abstract number: C5
Keywords: Bioengineering, Co-stimulation, Kidney, T cell activation
Session Information
Session Name: Poster Session C: Antigen Presenting Cells in Alloimmune Responses/B Cells and Antibody in Alloimmune Responses
Session Type: Poster Session
Date: Monday, May 4, 2015
Session Time: 5:30pm-6:30pm
 Presentation Time: 5:30pm-6:30pm
Presentation Time: 5:30pm-6:30pm
Location: Exhibit Hall E
Methods for preserving organs have employed hypothermic conditions. At these temperatures metabolism is 4% of normal. Tissue engineering of organs cannot be done without significant metabolism. Newer technologies only maintain organs at normothermic temperature for several hours. Such short periods limit the ability to adequately treat organs. We demonstrate the ability to tissue engineer renal allografts for costimlulatory blockade during 24 hours of warm (32C) acellular perfusion.
Methods – EMS acellular perfusion technology was used for tissue engineering. Human renal allografts (n=6) were placed on EMS perfusion. Upon restored metabolism the passenger leukocytes (PL) trapped within the renal parenchyma migrated into the perfusate. The PL were collected, phenotyped as CD209+ and treated with antibodies to CD80, CD86 and CD40. Costimulation was evaluated using a standard Brdu proliferation assay.
Results – Histology showed the migration of PL from the parenchyma mimicking the in vivo process that occurs following transplant. A reduction in the number of resident PL was observed within the kidneys with migration into the perfusate (12*10^6 cells). Since the perfusion is acellular, any cells found in the perfusate are of renal origin. Treating the PL with antibodies prevented a proliferative response demonstrating the potential for costimulatory blockade. 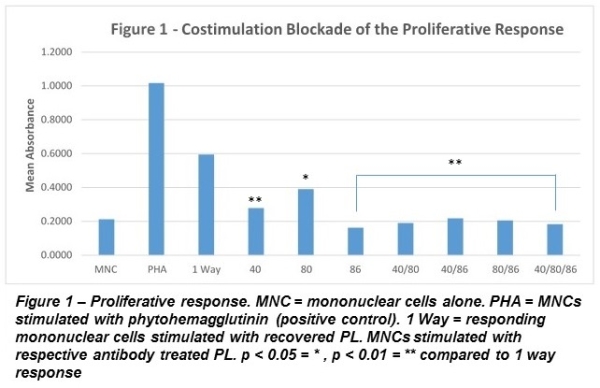
Conclusions – While the use of antibodies/fusion proteins for costimulation blockade have potential to prevent allograft rejection, these therapies are associated with severe complications. Costimulatory blockade during ex vivo perfusion, rather than systemic administration, could eliminate systemic side-effects, prevent direct antigen presentation and impact outcomes. Costimulatory molecules are also expressed on vascular endothelial cells and parenchymal cells. The vascular endothelium is a primary target of immune responses, strongly expresses CD86 along with MHC class II and functions as APCs. We are now testing the feasibility of administering antibody directly to the allograft. The ability to treat an allograft ex vivo could make multi-drug therapy feasible while minimizing side effects.
To cite this abstract in AMA style:
Brasile L, Henry N, Meyer A, Stubenitsky B. Tissue Engineering Human Kidneys for Costimulation Blockade [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/tissue-engineering-human-kidneys-for-costimulation-blockade/. Accessed May 17, 2026.« Back to 2015 American Transplant Congress
