The Fibrinogen-Like Protein 2 Molecule Influences the Development of Thymic Regulatory T-Cells
1Latner Thoracic Surgery Research Laboratories, University Health Network, Toronto, ON, Canada, 2Multi Organ Transplant Program, University Health Network, Toronto, ON, Canada, 3Toronto General Hospital Research Institute (TGHRI), University Health Network, Toronto, ON, Canada, 4Toronto General Hospital Research Institute, University Health Network, Toronto, ON, Canada
Meeting: 2020 American Transplant Congress
Abstract number: B-384
Session Information
Session Name: Poster Session B: Tolerance / Immune Deviation
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
*Purpose: Transplantation is the primary treatment for patients with end-stage organ failure. However, long-term outcomes are limited by immunosuppressant toxicities and chronic graft rejection. Identifying factors involved in regulatory T cell (Treg, CD4+ CD25+ FOXP3+) development and commitment is required for realization of their therapeutic potential in solid organ transplantation. Fibrinogen-like protein 2 (FGL2)-deficient Tregs are functionally defective, so we tested the hypothesis that FGL2 is required for normal thymic Treg development.
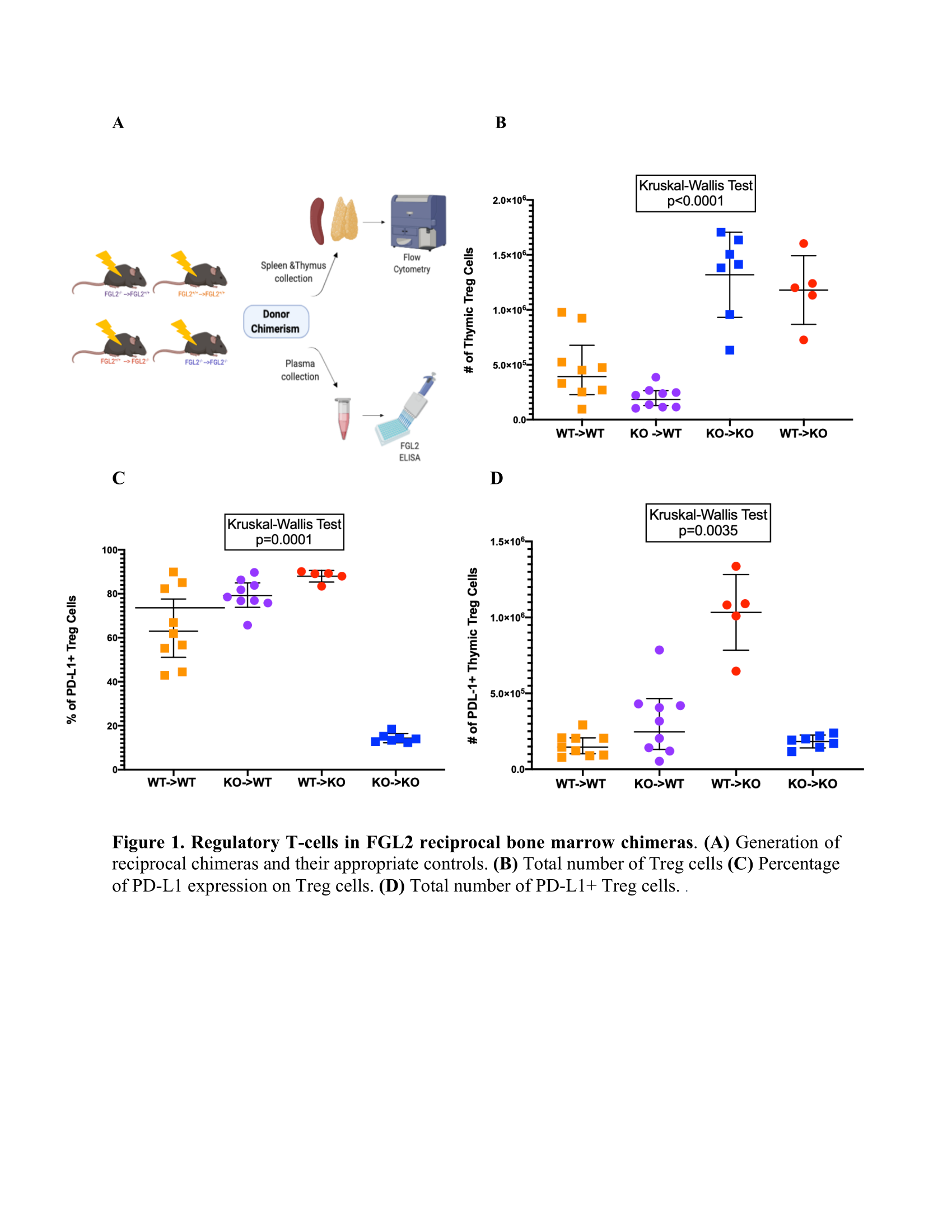
*Methods: We first generated reciprocal FGL2 bone marrow chimeras by reconstituting B6 CD45.1 or CD45.2 female mice with bone marrow stem cells from either FGL2-/- (KO) or FGL2+/+ (WT) mice, producing WT->KO and KO->WT chimeras and corresponding KO->KO and WT->WT controls (Fig.1A). Reconstitution was monitored by monthly tail bleed. Once the reconstitution reached ≥90%, plasma, spleens, and thymi were collected. We analyzed the plasma using ELISA for FGL2 and thymocytes with flow cytometry.
*Results: WT->KO chimeras had a faster rate of reconstitution than WT->WT chimeras. Analysis of thymocyte developmental stages revealed no significant differences between the groups. Thymi from KO->WT chimeras had a significantly lower number of donor-derived Tregs, whereas thymic Tregs were present in higher numbers in KO->KO and WT->KO chimeras (Fig.1B). PD-L1 expression differed dramatically among the groups, with KO->WT and WT->KO chimeras exhibiting an elevated percentage and absolute number of PD-L1+ cells compared with the other chimeras (Fig.1C-D). No differences were observed in GITR, TIGIT, LAG3, or PD-1 expression on thymic Tregs from the different groups. Lastly, there were no significant differences in FGL2 plasma levels between the different chimeras.
*Conclusions: Our results indicate that circulating FGL2 is derived from a radioresistant source and influences the rate of T cell reconstitution. Both radiosensitive and radioresistant sources of FGL2 affect thymic Treg expression of PD-L1, an important molecule for Treg function. In future work we will examine how FGL2 modulates Treg maturation, function and phenotype in the steady state and upon activation.
To cite this abstract in AMA style:
Lam C, Moshkelgosha S, Levy GA, Chruscinski A, Juvet SC. The Fibrinogen-Like Protein 2 Molecule Influences the Development of Thymic Regulatory T-Cells [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/the-fibrinogen-like-protein-2-molecule-influences-the-development-of-thymic-regulatory-t-cells/. Accessed June 3, 2026.« Back to 2020 American Transplant Congress