Population-Based Modeling of Prototypes and Determinants of Allograft Function Trajectories after Kidney Transplantation: Implications for Patient Monitoring and Risk Stratification
Paris Transplant Group, Paris, France.
Meeting: 2018 American Transplant Congress
Abstract number: 317
Keywords: Graft function, Kidney transplantation, Multicenter studies, Risk factors
Session Information
Session Name: Concurrent Session: Kidney Complications: Late Graft Failure
Session Type: Concurrent Session
Date: Monday, June 4, 2018
Session Time: 4:30pm-6:00pm
 Presentation Time: 4:42pm-4:54pm
Presentation Time: 4:42pm-4:54pm
Location: Room 6E
Background:
Although the current gold standard of monitoring kidney allograft patients relies primarily on GFR assessment, little is known about long-term eGFR trajectories prototypes and their determinants at a population level.
Methods:
An international, population-based cohort involving 10 transplant referral centers (7 in Europe and 3 in the US) was assembled with kidney transplants occurring from 2001 to 2016. Patients underwent assessment of clinical, histological, immunological and functional parameters including repeated eGFR measurements (MDRD). Latent class mixed models were fit to determine the prototypes of each patient's individual GFR trajectory. Multinomial regression models and PCA were used to assess transplant parameters that are associated with the distinct eGFR trajectories.
Results:
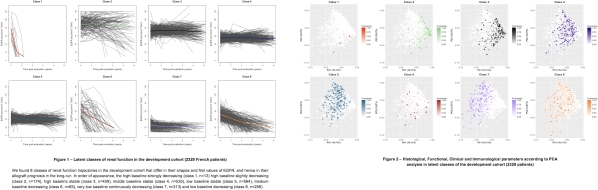
A total of 5,035 KTR were included. The median follow-up time was 6 years (IQR 4 – 9). A total of 53,024 eGFR measures were analyzed. Overall we identified 8 distinct latent class prototypes of eGFR trajectories (Figure 1). Multinomial regression and PCA analysis determined that recipient gender, expanded criteria donor, allograft inflammation (Banff i&t score), eGFR and proteinuria level at 1-year post-transplant, and circulating anti-HLA DSA, were the main determinants that discriminated between the latent eGFR classes (Figure 2). We confirmed that the eGFR trajectories, and their main clinical associations, were consistent in both the European and US cohorts.
Conclusion:
In this international, extensively-phenotypedpopulation ofKTR, we present novel insights into prototypes of individual eGFR trajectories and their main determinants. Our results provide the basis for using eGFR trajectory-based categorization of patients for improving patients risk stratification. These results also suggest the potential for a) early identification of adverse patient trajectories and the value of tailored therapeutic intervention, and b) the use of trajectory phenotypes in future clinical trials.
CITATION INFORMATION: Raynaud M., Aubert O., Naessens M., Morelon E., Giral M., Kamar N., Wong E., Jordan S., Orandi B., Segev D., Stegal M., Reese P., Lefaucheur C., Loupy A. Population-Based Modeling of Prototypes and Determinants of Allograft Function Trajectories after Kidney Transplantation: Implications for Patient Monitoring and Risk Stratification Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Raynaud M, Aubert O, Naessens M, Morelon E, Giral M, Kamar N, Wong E, Jordan S, Orandi B, Segev D, Stegal M, Reese P, Lefaucheur C, Loupy A. Population-Based Modeling of Prototypes and Determinants of Allograft Function Trajectories after Kidney Transplantation: Implications for Patient Monitoring and Risk Stratification [abstract]. https://atcmeetingabstracts.com/abstract/population-based-modeling-of-prototypes-and-determinants-of-allograft-function-trajectories-after-kidney-transplantation-implications-for-patient-monitoring-and-risk-stratification/. Accessed May 20, 2026.« Back to 2018 American Transplant Congress