Phenotype and Course of Renal Tubular Cell Death during Cold Ischemia versus Cold Ischemia Followed by Transplant
1Renal Division, University of Colorado, Aurora, CO, 2Transplant Microsurgery Program, University of Colorado, Aurora, CO
Meeting: 2019 American Transplant Congress
Abstract number: B9
Keywords: Apoptosis, Kidney transplantation
Session Information
Session Name: Poster Session B: Ischemia Reperfusion & Organ Rehabilition
Session Type: Poster Session
Date: Sunday, June 2, 2019
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall C & D
*Purpose: Treatments of Delayed graft function (DGF) are lacking. Prolonged cold ischemia (CI) is a risk factor for DGF but the pathways by which CI leads to DGF are unknown. The aim of this study was to determine the effects of CI on donor kidneys both during cold storage and after transplantation (CI+Txp). We hypothesized that CI alone would produce a different injury phenotype to CI+Txp.
*Methods: Male C57Bl/6J mice aged 8-12 weeks were subjected to either CI in saline at 4°C or to CI followed by syngeneic mouse kidney transplant. Unmanipulated kidneys served as controls. Renal tubular epithelial cell (RTEC) apoptosis, acute tubular necrosis (ATN) and renal function (serum creatinine) were quantitated. Proteins were examined by immunoblot or IHC.
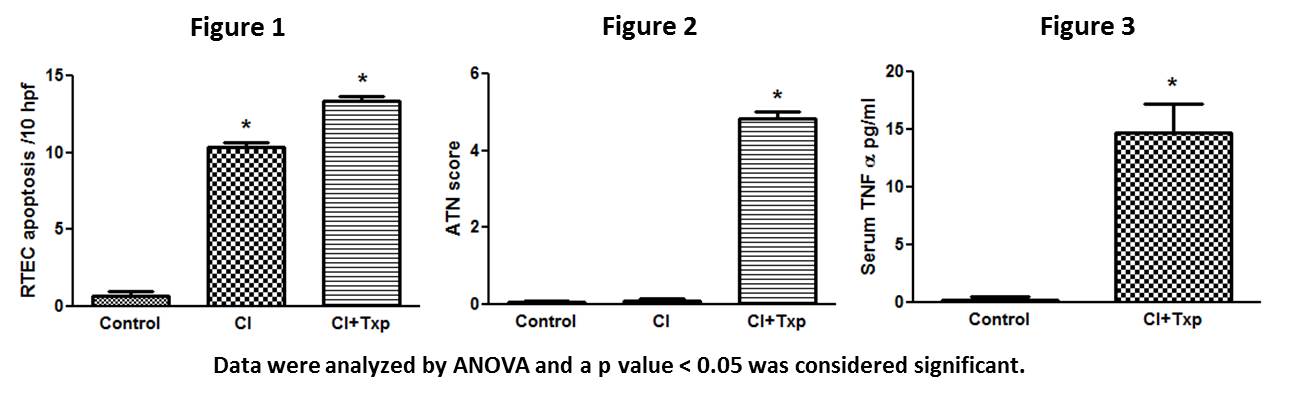
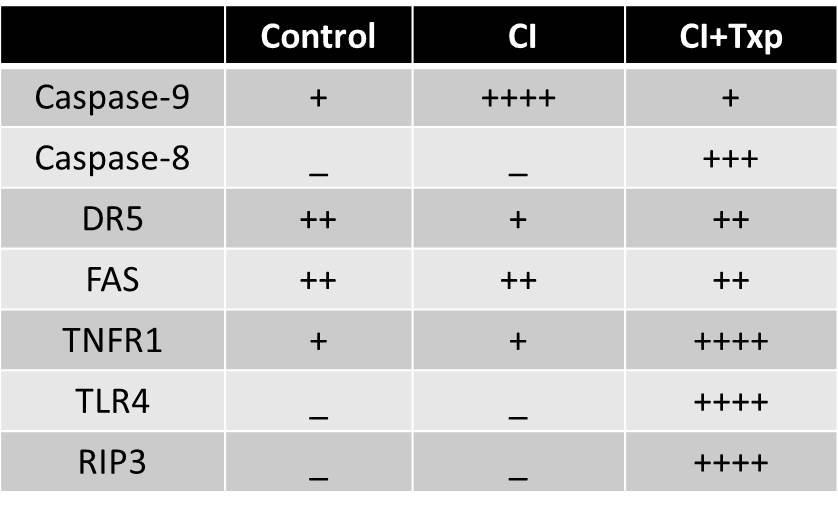
*Results: CI+Txp resulted in a significantly increased sCr (2.56±0.19) vs. controls (0.31±0.01). Kidneys exposed to CI alone demonstrated significantly increased (fig. 1) tubular cell apoptosis but no ATN (fig. 2) and increased active caspase-9 (see table). In contrast, CI+Txp led to (fig. 1) both tubular cell apoptosis and ATN (fig. 2) and increased death cleaved caspase-8 (see table). In addition, serum TNF was significantly elevated in CI+Txp vs controls (fig. 3). Expression of the upstream death receptors to caspase 8 revealed a significant increase in TNFR1 but no change in DR5 or Fas expression (see table). RTEC necrosis after CI+txp was associated with increased RIP3 and TLR4.
*Conclusions: CI results in RTEC apoptosis mediated by caspase-9, thus implicating the intrinsic pathway of apoptosis. Necrosis is not a feature of cold ischemic injury in this model. In contrast, CI+Txp results in a distinct injury phenotype of RTEC apoptosis mediated by death receptor pathway TNFR1 and caspase-8, thus implicating the extrinsic pathway of apoptosis. In addition, programmed necrosis was a feature of CI+Txp. Our data suggest prevention of apoptosis during cold storage might best be achieved by inhibition of the intrinsic pathway. In contrast, prevention of cell death after engraftment may require inhibition of both the extrinsic death receptor pathway via TNFR1 and inhibition of programmed necrosis via TLR4 and or TNFR1.
To cite this abstract in AMA style:
Jain S, Plenter R, Nydam T, Jani A. Phenotype and Course of Renal Tubular Cell Death during Cold Ischemia versus Cold Ischemia Followed by Transplant [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/phenotype-and-course-of-renal-tubular-cell-death-during-cold-ischemia-versus-cold-ischemia-followed-by-transplant/. Accessed May 16, 2026.« Back to 2019 American Transplant Congress