Optimizing the Immunogenicity and Immunomodulatory Properties of MSC for Cellular Therapy.
1ErasmusMC, Rotterdam, Netherlands
2Orbsen Therapeutics, Galway, Ireland
3University of Birmingham, Birmingham, United Kingdom
4Bioinvision, Mayfield Village.
Meeting: 2016 American Transplant Congress
Abstract number: D47
Keywords: Immune deviation, Immunogenicity, Inflammation, Stem cells
Session Information
Session Name: Poster Session D: Chimerism/Stem Cells, Cellular/Islet Transplantation, Innate Immunity, Chronic Rejection
Session Type: Poster Session
Date: Tuesday, June 14, 2016
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Halls C&D
Mesenchymal stromal cells (MSC) are candidates for immunotherapy after transplantation. MSC have shown to be immunomodulatory and low immunogenic, which is favorable for cellular therapy applicability, but there is evidence that MSC are more immunogenic than previously thought. Also, improving their immunomodulation would be useful. This study aims to optimize MSC by making them better immunomodulators and less immunogenic. Umbilical cord-derived MSC were treated for 3 days under various conditions, i.e. with pro/antiinflammatory cytokines, vitamins and serum-deprivation. Their immunogenicity and immunosuppressive capacity were examined by geneexpression analysis, surfacemarker expressions, IDOactivity and inhibition of Tcell proliferation. Next, susceptibility to NKlysis was investigated. Furthermore, treatment of liver inflammation and MSC survival was examined in a CCL4-induced liverdisease mousemodel. Results showed increased immunomodulatory capacity of MSC after IFNγ, IFNβ and TGFβ treatment. IFNγtreated MSC were the most potent inhibitors of Tcell proliferation as well as their IFNγ production. Furthermore, increased HLA I and II, were observed in IFNγ, IFNβ, vitB6 and Starv+VitB6treated MSC. These MSC were protected against NKlysis, which correlate to increased HLA I. In addition, TGFβMSC were protected against NKlysis even though HLA levels remained unchanged. Table 1 displays effects of the most prominent factors on immunogenicity and immunomodulatory capacity. In vivo, TGFβMSC improved persistence after infusion while clearance of Starv, VitB6 and Starv+VitB6treated MSC was accelerated compared to untreated MSC. Nonetheless, liverfunction showed a trend of improvement after administration of TGFβ and Starvtreated MSC. These data show versatility of MSC to culture conditions and possibility of optimizing MSC into less immunogenic cells with improved immunomodulatory properties, which is important for further development of MSC for immunotherapy.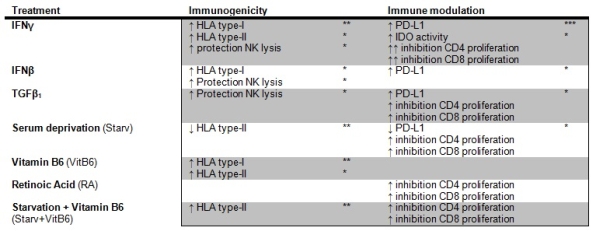
CITATION INFORMATION: de Witte S, Franquesa M, Strini T, Korevaar S, Luk F, Elliman S, Newsome P, Gargesha M, Roy D, Merino A, Baan C, Hoogduijn M. Optimizing the Immunogenicity and Immunomodulatory Properties of MSC for Cellular Therapy. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Witte Sde, Franquesa M, Strini T, Korevaar S, Luk F, Elliman S, Newsome P, Gargesha M, Roy D, Merino A, Baan C, Hoogduijn M. Optimizing the Immunogenicity and Immunomodulatory Properties of MSC for Cellular Therapy. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/optimizing-the-immunogenicity-and-immunomodulatory-properties-of-msc-for-cellular-therapy/. Accessed May 15, 2026.« Back to 2016 American Transplant Congress
