Long-Term Nonhuman Primate Renal Allograft Survival without Ongoing Immunosuppression in Recipients of Delayed Donor Bone Marrow Transplantation
Center for Transplantation Sciences, Massachusetts General Hospital, Boston, MA.
Meeting: 2018 American Transplant Congress
Abstract number: A395
Keywords: Bone marrow transplantation, Kidney transplantation, Survival, Tolerance
Session Information
Session Name: Poster Session A: Tolerance / Immune Deviation
Session Type: Poster Session
Date: Saturday, June 2, 2018
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Hall 4EF
Background: We have previously reported induction of renal allograft tolerance in nonhuman primates following an initial post-transplant period of conventional immunosuppression (IS) using a nonmyeloablative conditioning that included anti-CD154 (aCD154) and anti-CD8 (aCD8) mAbs plus horse ATG (hATG) and donor bone marrow transplantation (DBMT). Since these reagents are not clinically available, the protocol has been revised to be applicable to human recipients of deceased donor allografts. Method: All recipients initially underwent kidney transplantation (KT) alone with conventional IS, and underwent conditioning and DBMT four month after KT. Group 1 recipients received total body irradiation (3Gy), thymic irradiation (7Gy), and hATG. After DBMT, aCD154 for 2 weeks and cyclosporine for one month were given after which no further IS was administered. In Group 2, aCD8 was added to the Group 1 regimen. In Group 3, hATG, aCD8, and aCD154 were replaced with rabbit-ATG and CTLA4Ig (belatacept). Results: In Group 1, rapid recovery of CD8+ effector memory T cells (EM) and central memory T cells (CM) was observed by day 12, along with minimal induction of mixed chimerism (MC). 4/5 recipients failed to achieve long-term survival. In Group 2, the duration of CD8+ EM and CM deletion was extended to more than 30 days, and 10/12 developed MC. However only 6/12 achieved long-term survival because of death due to viral infection and PTLD. In Group 3, the deletion of CD8+ EM and CM was less intense than that observed in Group 2, but deletion of CD8+ EM was superior to that in Group 1 until day 20. This regimen, nevertheless, provided successful induction of MC in four consecutive recipients. Three recipients achieved long-term survival without infection or PTLD (one died due to anesthesia complication at the biopsy without rejection). Conclusion: This study provides proof of principle that induction of MC and long-term renal allograft survival without IS after delayed DBMT is possible with clinically available reagents.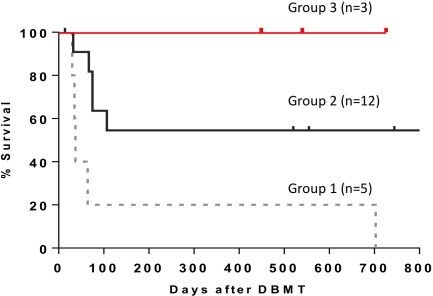
CITATION INFORMATION: Hotta K., Oura T., Dehnadi A., Matsunami M., Rosales I., Smith R., Colvin R., Cosimi A., Kawai T. Long-Term Nonhuman Primate Renal Allograft Survival without Ongoing Immunosuppression in Recipients of Delayed Donor Bone Marrow Transplantation Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Hotta K, Oura T, Dehnadi A, Matsunami M, Rosales I, Smith R, Colvin R, Cosimi A, Kawai T. Long-Term Nonhuman Primate Renal Allograft Survival without Ongoing Immunosuppression in Recipients of Delayed Donor Bone Marrow Transplantation [abstract]. https://atcmeetingabstracts.com/abstract/long-term-nonhuman-primate-renal-allograft-survival-without-ongoing-immunosuppression-in-recipients-of-delayed-donor-bone-marrow-transplantation/. Accessed May 20, 2026.« Back to 2018 American Transplant Congress
