Kidney Transplantation in Patients with Hereditary Kidney Diseases
1Saint Louis University, Saint Louis, MO, 2Division of Nephrology, Istanbul University, Istanbul, Turkey, 3Division of Nephrology, Uludag University, Bursa, Turkey, 4Department of Pathology, Istanbul University, Istanbul, Turkey
Meeting: 2021 American Transplant Congress
Abstract number: 961
Keywords: Genomics, Graft survival, Kidney transplantation, Living donor
Topic: Clinical Science » Kidney » Kidney Living Donor: Other
Session Information
Session Name: Kidney Living Donor: Other
Session Type: Poster Abstract
Session Date & Time: None. Available on demand.
Location: Virtual
*Purpose: Genetic testing is poised to play a larger role in the evaluation and management of both kidney transplant (KTx) recipients and donors. We characterized genetic variants and examined KTx outcomes among recipients with hereditary kidney diseases.
*Methods: A total of 96 prevalent KTx recipients with a blood relative diagnosed with a kidney disease by kidney biopsy, imaging or genetic testing were evaluated. Genetic sequencing, either whole exome sequencing (WES) (n=38) or a targeted gene panel (n=58), was performed. Graft loss risk was compared by donor type using multivariate Cox regression. Living donors were also followed for renal outcomes.
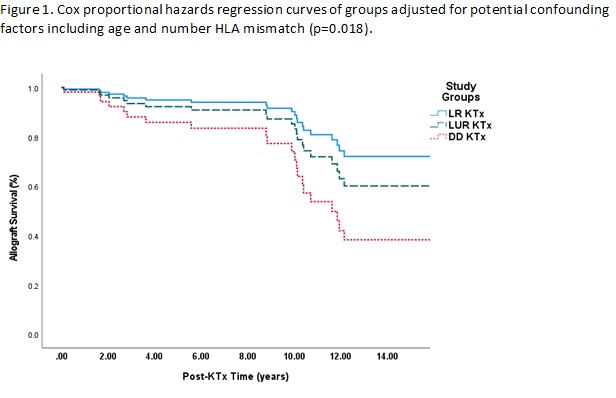
*Results: The genetically sequenced cohort comprised 58% men with mean age at KTx of 33±11; 70% were living related (LR), 8% living unrelated (LUR), and 22% deceased donor (DD) KTx recipients. We identified a diagnostic variant in 86% KTx recipients with hereditary kidney diseases including familial Mediterranean fever [43 of 43 recipients (100%)], FSGS (19/22, 86%), CAKUT (1/8, 12.5%), atypical hemolytic uremic syndrome (6/6, 100%), Alport syndrome (5/5, 100%), Fabry disease (4/4, 100%), C3 Glomerulopathy (4/4, 100%) and nephronophthisis (1/1, 100%). When grouped according to donor type, LUR KTx recipients were significantly older (48±7 yrs) than the LR (32±11 yrs) and DD (33±10 yrs) KTx recipients (p<0.001). Number of HLA mismatch was also higher in LUR KTx (4.1±0.8) than LR (2.8±1.1) and DD (3.0±0.9) KTx recipients (p<0.003). All groups were similar regarding gender, primary disease, post-KTx follow-up time. During a median follow up time of 9 years (IQR, 5-12), the rate of graft loss was significantly higher in DD KTx recipients [9/21 (43%)] compared to LUR [2/8 (25%)] and LR [11/67 (16%)] KTx groups (p=0.04). In Cox regression including adjustment for age and number HLA mismatches, DD KTx was significantly associated with 2.9-times increased risk of allograft loss (adjusted hazard ratio, 1.202.937.12) (p=0.018). Of 67 LR donors, only one (1.5%) developed nephrotic range proteinuria and biopsy confirmed FSGS during 9 years of follow-up. None of the LR donors required renal replacement treatment.
*Conclusions: Living donor KTx is the best option for ESKD patients with hereditary kidney diseases. Although the risk is low, the possible occurrence of the same disease in the related living donor can happen.
To cite this abstract in AMA style:
Caliskan Y, Garayeva N, Dirim AB, Safak S, Mirioglu S, Yazici H, Yildiz A, Oto OA, Demir E, Ozluk Y, Turkmen A, Lentine K. Kidney Transplantation in Patients with Hereditary Kidney Diseases [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/kidney-transplantation-in-patients-with-hereditary-kidney-diseases/. Accessed May 19, 2026.« Back to 2021 American Transplant Congress