Investigating the HLA Alloimmune Background of the Histological Changes Suggestive of Antibody-mediated Injury in the Absence of Donor-specific Anti-HLA Antibodies
Microbiology, Immunology and Transplantation, KU Leuven, Leuven, Belgium
Meeting: 2021 American Transplant Congress
Abstract number: 454
Keywords: Histocompatibility antigens, HLA antibodies, Kidney transplantation, Rejection
Topic: Clinical Science » Kidney » Kidney Acute Antibody Mediated Rejection
Session Information
Session Name: Kidney Alloimmune Responses
Session Type: Poster Video Chat
Date: Monday, June 7, 2021
Session Time: 7:30pm-8:30pm
 Presentation Time: 8:10pm-8:20pm
Presentation Time: 8:10pm-8:20pm
Location: Virtual
*Purpose: The histology of antibody-mediated rejection is observed frequently after kidney transplantation, but a significant percentage of the patients do not have detectable donor-specific HLA antibodies (DSAnegABMRh). Although part of these cases could be related to the presence of non-HLA antibodies, until now, none of the previously suggested non-HLA antibodies are routinely tested in clinical practice for the diagnosis of ABMR. While there is an active interest in non-HLA antibodies’ role to DSAnegABMRh, it remains crucial to exclude the possible contribution of the HLA incompatibility to this phenotype. We aimed to investigate the associations between the HLA molecular mismatches and the occurrence of the DSAnegABMRh phenotype.
*Methods: All consecutive kidney recipients transplanted at a single center between 2004 and 2013 were eligible for this study. The transplant pairs were genotyped at high-resolution for 11 HLA loci. HLAMatchmaker, PIRCHE-II and HLA-EMMA tools were used to determine the number of HLA molecular mismatches (MM).
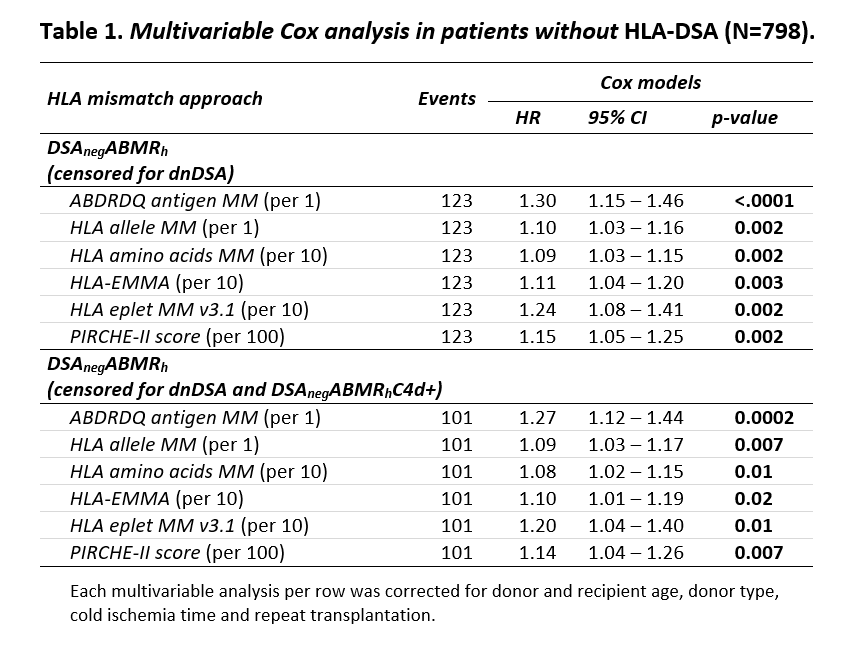
*Results: Patients with pretransplant donor-specific HLA antibodies were excluded from this study. Of the remaining 798 kidney transplant recipients with available biopsy follow-up, 123 (15.4%) developed ABMRh in the absence of de novo HLA-DSA. In adjusted multivariable Cox analysis, HLA antigen (HR=1.30 per 1; 95%CI 1.15-1.46; p<.0001), HLA allele (HR=1.10 per 1; 95%CI 1.03-1.16; p=0.002) and HLA amino acid mismatches (HR=1.09 per 10; 95%CI 1.03-1.15; p=0.002) were identified as risk factors for developing DSAnegABMRh. Similarly, all the different HLA molecular mismatches, HLA-EMMA (HR=1.11 per 10; 95%CI 1.04-1.20; p=0.003), eplet MM (HR=1.24 per 10; 95%CI 1.08-1.41; p=0.002) and PIRCHE-II (HR=1.15 per 100; 95%CI 1.05-1.25; p=0.002) associated with DSAnegABMRh. The subsequent multivariate Cox model, censored for cases with DSAnegABMRh and C4d deposition (ABMR according to Banff 2019), confirmed that HLA incompatibility is a risk factor for developing DSAnegABMRh. Finally, in a sensitivity analysis restricted to anti-HLA antibody-negative patients (n=660), again, all different levels of HLA mismatches were independently associated with the DSAnegABMRh rejection phenotype.
*Conclusions: HLA mismatches associate with DSAnegABMRh, also in patients without any sign of circulating HLA antibodies. This indicates that the donor-recipient HLA-incompatibility at least partially explains the development of the histological features of ABMR, in an HLA antibody-independent process.
To cite this abstract in AMA style:
Senev A, Naesens M. Investigating the HLA Alloimmune Background of the Histological Changes Suggestive of Antibody-mediated Injury in the Absence of Donor-specific Anti-HLA Antibodies [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/investigating-the-hla-alloimmune-background-of-the-histological-changes-suggestive-of-antibody-mediated-injury-in-the-absence-of-donor-specific-anti-hla-antibodies/. Accessed May 20, 2026.« Back to 2021 American Transplant Congress