Insulin Treatment in Pancreas Donors on the Intensive Care is a Marker of Beta Cell Death as Evidenced by microRNA Data
University of Manchester, Manchester, United Kingdom
Meeting: 2019 American Transplant Congress
Abstract number: A128
Session Information
Session Name: Poster Session A: Islet Cell and Cell Transplantation
Session Type: Poster Session
Date: Saturday, June 1, 2019
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Hall C & D
*Purpose: We showed in prior studies that donor insulin use on intensive care predicts early beta cell dysfunction in islet and solid pancreas transplantation. We hypothesized that beta cell death explains this requirement for insulin.
*Methods: Plasma samples from 100 brain dead pancreas donors were collected by the Quality in Organ Donation Biobank: a) at consent; and b) at the time of aortic cross-clamp. Beta cell death was assessed from expression of miR34a (pro-apoptotic), miR21 (anti-apoptotic) and miR-375 (beta-cell death marker). Immunoassays assessed levels of circulating inflammatory cytokines and insulin secretion. We compared biomarker levels between insulin-treated and non-insulin-treated donors (ITD vs NITD) and between time-points within-groups.
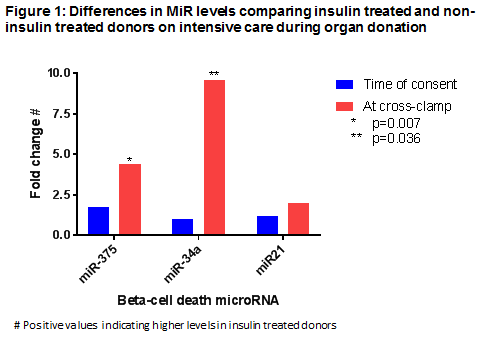
*Results: Insulin was received by 43 (43%) donors; there were no significant differences in clinical variables by insulin use. C-peptide levels across the 2 time points were lower in in ITD vs NITD (63 [26-304] vs 296 [45 vs 181] ng.hour/mL, p=0.001). In ITD vs NITD, at the time of retrieval, MiR-375 levels were 4-fold higher (p=0.007), miR-34a 10-fold higher (p=0.036) and miR-21 was similar (numerically 2-fold higher, p=0.243; figure 1). In NITD, between consent and cross clamp, mir-375 levels were reduced 6-fold (p=0.050), miR21 reduced 4-fold (p=0.016) and miR-34a levels were unchanged (numerically 3-fold lower; p=0.465), whereas in ITD, all 3 miR levels were unchanged. IL-6 levels were lower in ITD vs NITD at the time of consent (43 [16-100] vs 154 [33-633] pg/mL, p=0.001) and cross-clamp (54 [30-109] vs 117 [47-222] pg/mL, p=0.005).
*Conclusions: In ITD, greater expression of beta cell death-associated microRNA and lower IL-6 levels provide a possible mechanistic explanation for post-transplant beta-cell dysfunction in prior studies. In NITD, time-related reduction of microRNAs suggests lowered activity in pathophysiological pathways leading to beta cell death. These microRNAs provide potential therapeutic targets; intervention studies could determine if this approach could improve transplant outcomes.
To cite this abstract in AMA style:
Shapey I, Summers A, O'Sullivan J, Yiannoullou P, Hanley N, Augustine T, Rutter M, Dellen Dvan. Insulin Treatment in Pancreas Donors on the Intensive Care is a Marker of Beta Cell Death as Evidenced by microRNA Data [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/insulin-treatment-in-pancreas-donors-on-the-intensive-care-is-a-marker-of-beta-cell-death-as-evidenced-by-microrna-data/. Accessed May 19, 2026.« Back to 2019 American Transplant Congress