Influence of Donor-Specific Antibodies on Renal Function in Kidney Transplant Recipients Following Early Switch from Calcineurin Inhibitor to Everolimus: Subanalysis from the ELEVATE Study.
1ELEVATE Study Group, Llobregat Catalunya, Spain
2Novartis Pharma AG, Basel, Switzerland
Meeting: 2017 American Transplant Congress
Abstract number: 172
Keywords: Graft function, HLA antibodies, Kidney transplantation, Renal function
Session Information
Session Name: Concurrent Session: Novel Immunosuppression - DSA Monitoring
Session Type: Concurrent Session
Date: Sunday, April 30, 2017
Session Time: 4:30pm-6:00pm
 Presentation Time: 5:42pm-5:54pm
Presentation Time: 5:42pm-5:54pm
Location: E450b
Purpose: The development of donor-specific antibody (DSA) is a known risk factor for poor graft outcomes after kidney transplantation (KTx). Previous analysis of the ELEVATE (NCT01114529) study showed the effect of DSA on treated biopsy-proven acute rejection and antibody-mediated rejection in KTx recipients (KTxRs). Here, we compare the renal function outcomes by the presence of preformed and de novo DSA in KTxRs receiving everolimus (EVR) vs standard calcineurin inhibitor (CNI).
Methods: ELEVATE was a 24-month (M), multicenter, open-label study, in which de novo KTxRs were randomized at 10-14 weeks post-KTx to receive EVR (n=360; C0:6-10 ng/mL) or continue CNI regimen (n=357; tacrolimus [TAC] C0: 5-10 ng/mL; cyclosporine A [CsA] C0: 100-250 ng/mL). Preformed DSA was defined as a mean fluorescence intensity (MFI) value ≥500 at randomization (RND), whereas de novo DSA was defined by MFI ≥500 at any time post-RND in patients with MFI <500 at RND. Evolution of renal function (estimated glomerular filtration rate [eGFR; MDRD4]), proteinuria, and serum creatinine were assessed from RND to M24.
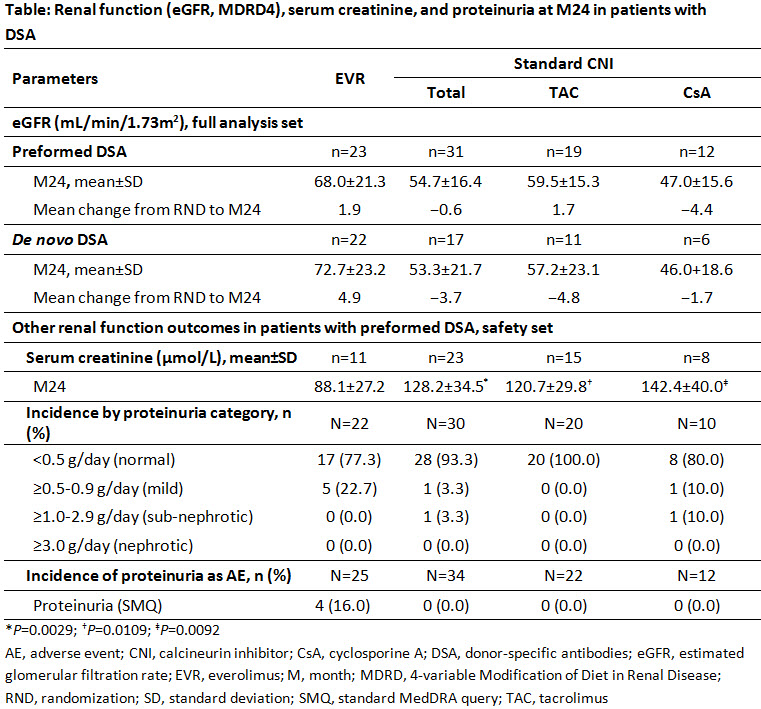
Results: Overall, 23 and 31 patients in the EVR and CNI arms with preformed DSA, and 22 and 17 patients with de novo DSA were available for the analysis of renal function at M24, respectively. At M24, eGFR was numerically higher in EVR vs CNI arm irrespective of the presence of preformed (68.0 vs 54.7 mL/min/1.73m2) or de novo (72.7 vs 53.3 mL/min/1.73m2) DSA (Table). In patients with preformed DSA, the mean creatinine level was significantly lower in EVR vs CNI arm (P=0.0029). No patients had sub-nephrotic/nephrotic proteinuria in EVR arm. Incidence of proteinuria as an adverse event was observed in four patients in EVR, and none in CNI arm (Table).
Conclusion: The findings of current analysis, though limited by sample size, suggest that EVR offers long-term preservation of renal function vs CNI irrespective of the presence of preformed or de novo DSA.
CITATION INFORMATION: Cruzado J, Holdaas H, Lopez P, Bernhardt P, Bader G, Claas F, Arns W, van der Giet M, de Fijter J. Influence of Donor-Specific Antibodies on Renal Function in Kidney Transplant Recipients Following Early Switch from Calcineurin Inhibitor to Everolimus: Subanalysis from the ELEVATE Study. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Cruzado J, Holdaas H, Lopez P, Bernhardt P, Bader G, Claas F, Arns W, Giet Mvander, Fijter Jde. Influence of Donor-Specific Antibodies on Renal Function in Kidney Transplant Recipients Following Early Switch from Calcineurin Inhibitor to Everolimus: Subanalysis from the ELEVATE Study. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/influence-of-donor-specific-antibodies-on-renal-function-in-kidney-transplant-recipients-following-early-switch-from-calcineurin-inhibitor-to-everolimus-subanalysis-from-the-elevate-study/. Accessed May 28, 2026.« Back to 2017 American Transplant Congress