Ideal Versus Total Body Weight Dosing of Rabbit Anti-Thymocyte Globulin Induction Therapy on Patient Outcomes in High Immunological Risk Kidney Transplant
Duke University Hospital, Durham, NC.
Meeting: 2015 American Transplant Congress
Abstract number: A151
Keywords: Antilymphocyte antibodies, Dosage
Session Information
Session Name: Poster Session A: Kidney Immunosuppression: Induction Therapy
Session Type: Poster Session
Date: Saturday, May 2, 2015
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Exhibit Hall E
Background
The optimal dose of rabbit anti-thymocyte globulin (rATG) induction therapy in high immunological risk kidney transplantation (KTx) lacks consensus and the effect of dosing weight has not been evaluated. Before 2009, high immunologic risk KTx recipients, defined as panel reactive antibody greater than 40%, 2nd KTx with early loss, or 3rd and greater KTx, received a 5-day total rATG dose of 7.5 mg/kg using total body weight (TBW). In 2009, this dosing strategy was modified to utilize ideal body weight (IBW).
Methods and materials
This retrospective, single-center cohort study included all adult high immunologic risk KTx recipients from 1/1/2006 to 10/31/2012 who received 5 doses of rATG for induction therapy, excluding those who had a dose reduction due to leukopenia or thrombocytopenia. All patients were discharged on tacrolimus, mycophenolate, and prednisone. Clinical outcomes were evaluated between the TBW and IBW dosing strategies by using an a priori proxy marker of cumulative dose of 7.5 mg/kg TBW to separate patients into two groups. The primary endpoint was rate of first biopsy-proven acute rejection (BPAR) within the first 90 days post-KTx.
Results
Eighty-nine patients were included. Baseline demographics were similar between groups except for mean TBW was significantly higher in the IBW cohort (86.4 kg vs. 75.7 kg; p = 0.009) and cumulative rATG dose per TBW was significantly less for the IBW cohort (6.7 mg/kg vs. 8.1 mg/kg TBW; p < 0.001). Rate of BPAR at 90-days and 1-year were similar between IBW and TBW cohorts (90-days, 4.2% vs. 0%, p =0.5; 1-year, 8.7% vs. 0%, p = 0.13). Patient survival and renal function were also similar. Median cost of rATG therapy per patient was less in the IBW cohort; however, this was not statistically significant ($17,542 vs. $19,934; p = 0.3).
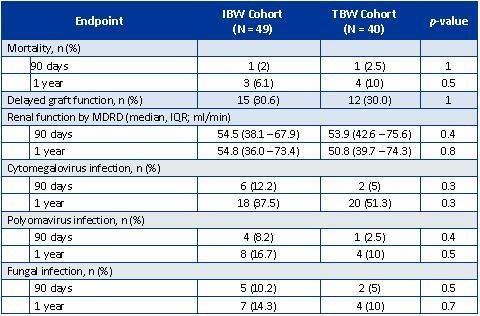
Conclusion
Our results suggest that use of IBW for dose calculations of rATG induction therapy in high immunological risk KTx at 7.5 mg/kg provides low rates of acute rejection with a similar safety profile as that of a TBW-dosed regimen. Use of IBW for lower cumulative doses may still need further evaluation.
To cite this abstract in AMA style:
Vacha M, Gommer J, Rege A, Sanoff S, Sudan D, Harris M. Ideal Versus Total Body Weight Dosing of Rabbit Anti-Thymocyte Globulin Induction Therapy on Patient Outcomes in High Immunological Risk Kidney Transplant [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/ideal-versus-total-body-weight-dosing-of-rabbit-anti-thymocyte-globulin-induction-therapy-on-patient-outcomes-in-high-immunological-risk-kidney-transplant/. Accessed May 15, 2026.« Back to 2015 American Transplant Congress
