Hepatocyte SIRT1 Suppresses Atf4-mediated Apoptosis Triggered by Cold Stress in Mouse and Human Liver Transplantation
1Dumont-UCLA Transplant Center, Los Angeles, CA, 2NIEHS, Durham, NC
Meeting: 2021 American Transplant Congress
Abstract number: 354
Keywords: Hepatocytes, Liver preservation, Liver transplantation
Topic: Basic Science » Ischemia Reperfusion & Organ Rehabilitation
Session Information
Session Name: Ischemia Reperfusion & Organ Rehabilitation
Session Type: Rapid Fire Oral Abstract
Date: Tuesday, June 8, 2021
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:20pm-6:25pm
Presentation Time: 6:20pm-6:25pm
Location: Virtual
*Purpose: The role of ER-stress or hepatocyte-specific SIRT1 signaling during donor liver cold preservation remains unknown. We employed a mouse OLT model and analyzed clinical liver transplant biopsies (Bx) to determine as to whether and how hepatocyte SIRT1- ER stress (p-eIF2α/ATF4)-apoptosis axis may affect outcomes.
*Methods: WT or hepatocyte-specific SIRT1 deficient (hSIRT1-KO) livers (C57/BL6), subjected to extended (18h) cold storage (4C in UW solution), were transplanted to syngeneic WT recipients. Liver/serum samples were collected at 6h post-reperfusion. Human liver Bx collected from 60 OLT patients (after cold storage and 2 hrs after reperfusion) were analyzed by Western blots/RT-PCR.
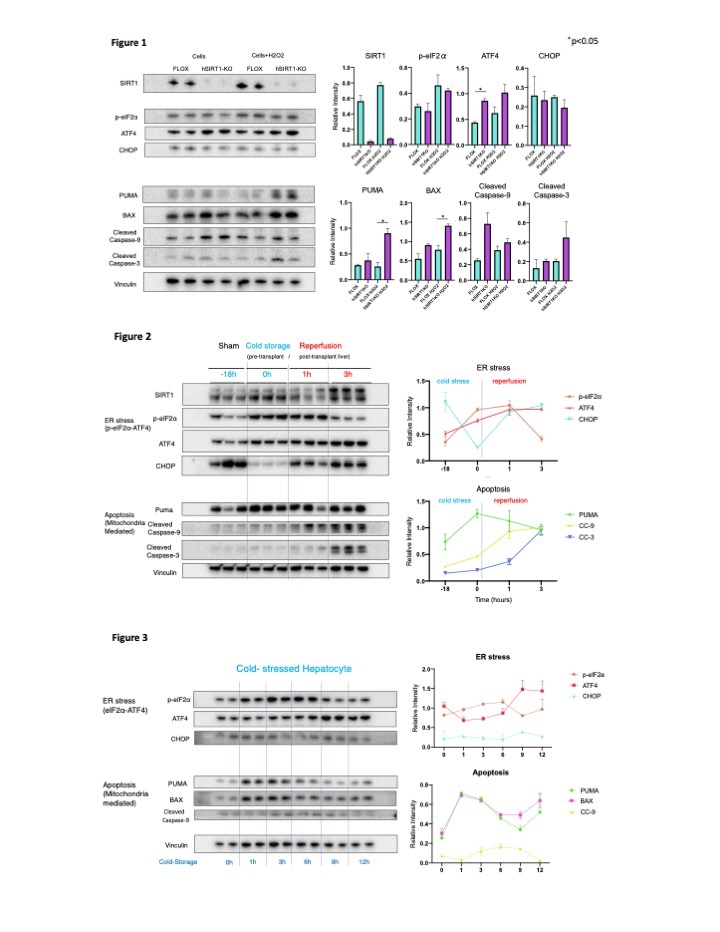
*Results: hSIRT1-KO primary hepatocytes showed increased levels of ATF4 and up-regulation of PUMA/BAX/Cleaved Caspase-9 while levels of p-eIF2α/CHOP remained unchanged as compared to WT counterparts (Figure 1). Furthermore, silencing (siRNA) of ATF4 reduced levels of PUMA/BAX but not of CHOP in hepatocyte cultures. Compared with WT liver grafts (WT>WT), disruption of hepatocyte SIRT1 signaling in the donor liver (hSIRT1-KO>WT) aggravated IRI, as evidenced by: 1/ sALT/sAST levels (ALT: 7321±6104 vs 3875±1869 IU/L, n=12/12, p=0.037; AST: 3671±2678 vs 1853±532 IU/L, n=12/12, p=0.015); 2/ Suzuki’s histological grading (p<0.05); 3/ frequency of TUNEL+ cells (p<0.05); 4/ augmented neutrophil/macrophage infiltration (IHC, p<0.05); 5/ increased hepatic ATF4/PUMA/Cleaved Caspase-9/Cleaved Caspase-3 expressions (WB, p<0.05); and 6/ impaired survival of IR-stressed OLTs (2-weeks: 11% vs 44%, n=9/9, p=0.036). Cold-stored liver grafts showed up-regulation of p-eIF2α/ATF4 and PUMA/Cleaved Caspase-9 while CHOP levels decreased (Figure 2). Consistently, cold-stressed hepatocytes displayed increased levels of p-eIF2α/ATF4 and PUMA/BAX/Cleaved Caspase-9, with CHOP relatively unchanged (Figure 3). In the clinical arm, pre-reperfusion SIRT1 levels in human liver Bx negatively correlated with protein expression levels of ATF4 (r=-0.313, p=0.056) but positively with Bcl-2 (r=0.4519, p=0.0003), while post-reperfusion hepatic SIRT levels in OLTs negatively correlated with ATF4 (r=-0.2684, p=0.0058) and Cleaved Caspase-3 (r=-0.4213, p=0.0111).
*Conclusions: This translational study documents hepatocyte SIRT1 regulates ATF4 mediated apoptosis triggered by cold-stress, providing a novel therapeutic target and putative mechanism of hepatocyte-specific cell death pathway.
To cite this abstract in AMA style:
Kadono K, Hirao H, Kojima H, Dery KJ, Aziz J, Li X, Kupiec-Weglinski JW. Hepatocyte SIRT1 Suppresses Atf4-mediated Apoptosis Triggered by Cold Stress in Mouse and Human Liver Transplantation [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/hepatocyte-sirt1-suppresses-atf4-mediated-apoptosis-triggered-by-cold-stress-in-mouse-and-human-liver-transplantation/. Accessed June 3, 2026.« Back to 2021 American Transplant Congress