End Stage Renal Disease after Face Transplant
1University of Maryland, Baltimore, MD
2Ochsner Health Center, Covington, LA
3New York University, New York, NY.
Meeting: 2018 American Transplant Congress
Abstract number: 287
Keywords: Calcineurin, Renal failure, Renal injury
Session Information
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:42pm-3:54pm
Presentation Time: 3:42pm-3:54pm
Location: Room 303
Purpose Renal failure as an outcome of chronic immunosuppression is a known complication of organ transplantation. We report the first case of end stage renal disease resulting from calcineurin inhibitor (CNI) based immunosuppression in vascularized composite allotransplantation (VCA).
Methods A full-face transplant was performed with maxilla, mandible, tongue, skin, and scalp on March 2012. Induction immunosuppression was provided by alemtuzumab and maintenance therapy with tacrolimus (Tac), mycophenolate mofetil, and prednisone. Tac target levels of 6-8 ng/mL during the first year were lowered to 5 ng/mL in subsequent years. The patient maintained regular nephrology and transplant follow up.
Results Three clinically significant graft rejection episodes occurred on postoperative days 28, 402, and 712 with Banff I, I, and II scores respectively. Episode 1 occurred after weaning steroids, episode 2 after sub-therapeutic Tac levels, and the most severe episode 3 after decreased Tac and introduction of everolimus as a renal protective strategy. After year 2 no further rejection episodes occurred with maintenance therapy MMF 500 mg bid, prednisone 5 mg daily, and a Tac target level 5 ng/mL. Creatinine preoperatively was noted to be 0.6-1.1 mg/dL. Year 2 average creatinine levels remained elevated at 2 mg/dL. Year 5 creatinine increased to 5 mg/dL with non-oliguric CKD Stage 5 and an eGFR of 15 mL/min/BSA. 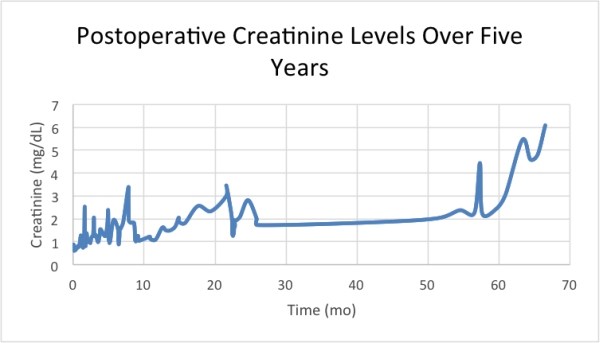 CT imaging revealed atrophy of native kidneys consistent with renal disease. The patient has been listed for renal transplantation. Biopsy at year 5 demonstrated no evidence of rejection or chronic vasculopathy. Clinical appearance and function remained excellent.
CT imaging revealed atrophy of native kidneys consistent with renal disease. The patient has been listed for renal transplantation. Biopsy at year 5 demonstrated no evidence of rejection or chronic vasculopathy. Clinical appearance and function remained excellent.
Conclusions This is the first reported case of ESRD as a direct consequence of immunosuppression in VCA. Understanding complications of VCA with standard immunosuppression is critical in risk/benefit considerations for VCA candidates. Continued research into alternate immunosuppressive approaches to achieve excellent graft outcomes with minimal complications is critical to the future success of this field.
CITATION INFORMATION: Shockcor N., Buckingham B., Hassanein W., Haririan A., Roberts S., Nam A., Rodriguez E., Bartlett S., Barth R. End Stage Renal Disease after Face Transplant Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Shockcor N, Buckingham B, Hassanein W, Haririan A, Roberts S, Nam A, Rodriguez E, Bartlett S, Barth R. End Stage Renal Disease after Face Transplant [abstract]. https://atcmeetingabstracts.com/abstract/end-stage-renal-disease-after-face-transplant/. Accessed May 11, 2026.« Back to 2018 American Transplant Congress
