Costimulation Blockade Modulates Humoral Responses in Highly Sensitized Transplant Recipients
Surgery, Duke Transplant Center, Durham, NC
Meeting: 2019 American Transplant Congress
Abstract number: 26
Keywords: Alloantibodies, B cells, Co-stimulation, T helper cells
Session Information
Session Name: Concurrent Session: Kidney Immunosuppression: Desensitization
Session Type: Concurrent Session
Date: Sunday, June 2, 2019
Session Time: 2:30pm-4:00pm
 Presentation Time: 2:30pm-2:42pm
Presentation Time: 2:30pm-2:42pm
Location: Ballroom A
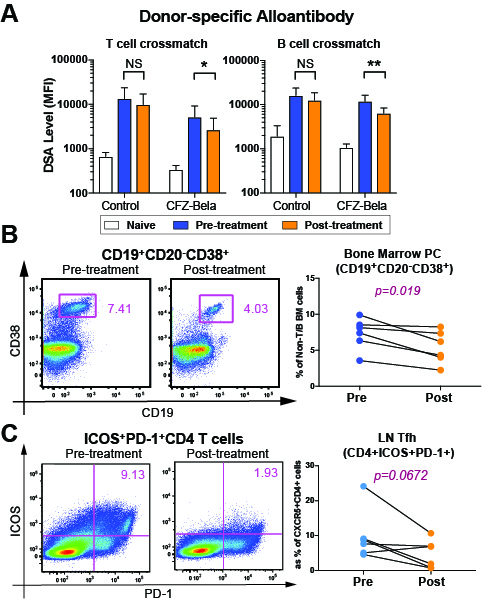
*Purpose: Broad human leukocyte antigen sensitization is a significant barrier to successful solid organ transplantation. Sensitized patients have poor access to donor organs, high wait list mortality, and when transplanted, inferior graft survival due to acute and chronic antibody-mediated injuries. We hypothesized that simultaneously targeting germinal center (GC) response with belatacept (Bela) and plasma cells (PC) with carfilzomib (CFZ) would produce durable desensitization without interfering protective humoral immunity in highly sensitized nonhuman primate kidney transplantation.
*Methods: Maximally MHC-mismatched rhesus pairs were sensitized with two sequential skin transplants and then desensitized with CFZ (27 mg/m2 IV) and Bela (20 mg/kg IV) weekly for one month. Blood, lymph node (LN), and bone marrow (BM) cells as well as serum were analyzed pre-, post-desensitization, and post-kidney transplantation.
*Results: CFZ/Bela (n=6) significantly reduced donor-specific antibody (DSA) (p = 0.0453). However, anti-tetanus and anti-CMV (gB) antibodies were not affected by the treatment. While circulating T and B cell counts were unchanged, CFZ/Bela significantly reduced BM-PC (CD19+CD38+, 7.3±2.1 vs. 5.4±2.2; p=0.019) and showed a strong trend of reduced LN-Tfh cells (CD4+ICOS+PD1hi , 9.9±7.2 vs. 6.9±4.6; p=0.06) (Fig 1). Unexpectedly, LN Treg cells also declined (CD4+CD25+CD127lo, 11.8±2.4 vs. 8.2±1.6; p=0.038). However, LN-Tfr cells were not significantly reduced (CD4+CXCR5+ICOS+PD1hi; p=0.13). Graft survival was significantly prolonged (p=0.016) and renal biopsy at one month showed significant reduction of AMR score (g+ptc+c4d; p=0.012) in CFZ/Bela treated animals compared to controls (n=4). Furthermore, all animals showed no CMV reactivation as was seen in parallel experiments using dual costimulation blockade with bortezomib. However, four out of five long-term animals showed gradual rebound of LN-PC and LN-Tfh cells while BM-PC did not fully recover by 100 days.
*Conclusions: We employed a mechanistically designed desensitization regimen using proteasome inhibitor and costimulation blockade in a rigorous, highly sensitized nonhuman primate model and demonstrate that this regimen reduces DSA, BM-PCs, and LN-Tfh cells without reducing anti-TT and anti-CMV Ab levels. Even though desensitization with CFZ/Bela markedly prolongs allograft survival, most animals eventually experienced AMR with rebound GC response, suggesting that desensitization needs to be maintained using ongoing suppression of the B cell response.
To cite this abstract in AMA style:
Kwun J, Ezekian B, Schroder P, Manook M, Yoon J, Mulvihill M, Freischlag K, Yi J, Knechtle S. Costimulation Blockade Modulates Humoral Responses in Highly Sensitized Transplant Recipients [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/costimulation-blockade-modulates-humoral-responses-in-highly-sensitized-transplant-recipients/. Accessed May 15, 2026.« Back to 2019 American Transplant Congress