Combined Antithymocyte Globulin (ATG) and Rituximab (RTX) Induction in African American (AA) Kidney Transplant Recipients
Methodist Hospital, Houston, TX
Meeting: 2019 American Transplant Congress
Abstract number: B198
Keywords: B cells, Immunosuppression, Induction therapy, Kidney transplantation
Session Information
Session Name: Poster Session B: Kidney Immunosuppression: Induction Therapy
Session Type: Poster Session
Date: Sunday, June 2, 2019
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall C & D
*Purpose: We have previously shown that despite the use of T cell-depleting antibody induction, AA kidney transplant recipients (KTR) remain at high risk of rejection and de novo donor specific antibodies (dnDSA) (Patel SJ et al, Transpl Int 2016:29(8);897). Thus, we sought to examine the safety and efficacy of combined B and T cell-depleting induction using RTX with ATG in AA KTR.
*Methods: This was a single-center, retrospective review of two cohorts of sensitized (PRA>20%) but crossmatch-negative deceased donor AA KTR. From 01/2014 to 01/2015, patients received ATG induction alone (4.5-6 mg/kg over 3-4 days; “ATG” group). Thereafter, a second cohort began receiving 1 dose of RTX 375 mg/m2 in addition to ATG (“ATG+RTX” group). Per protocol, all patients underwent prospective dnDSA screening with renal biopsy in the setting of positive dnDSA, as well as screening for viral replication (CMV, BK, JC, and EBV). All KTR received tacrolimus, mycophenolate, and prednisone. Graft outcomes and rates of viral infections at 1 year were compared between ATG+RTX (n=48) and ATG alone (n=24) groups.
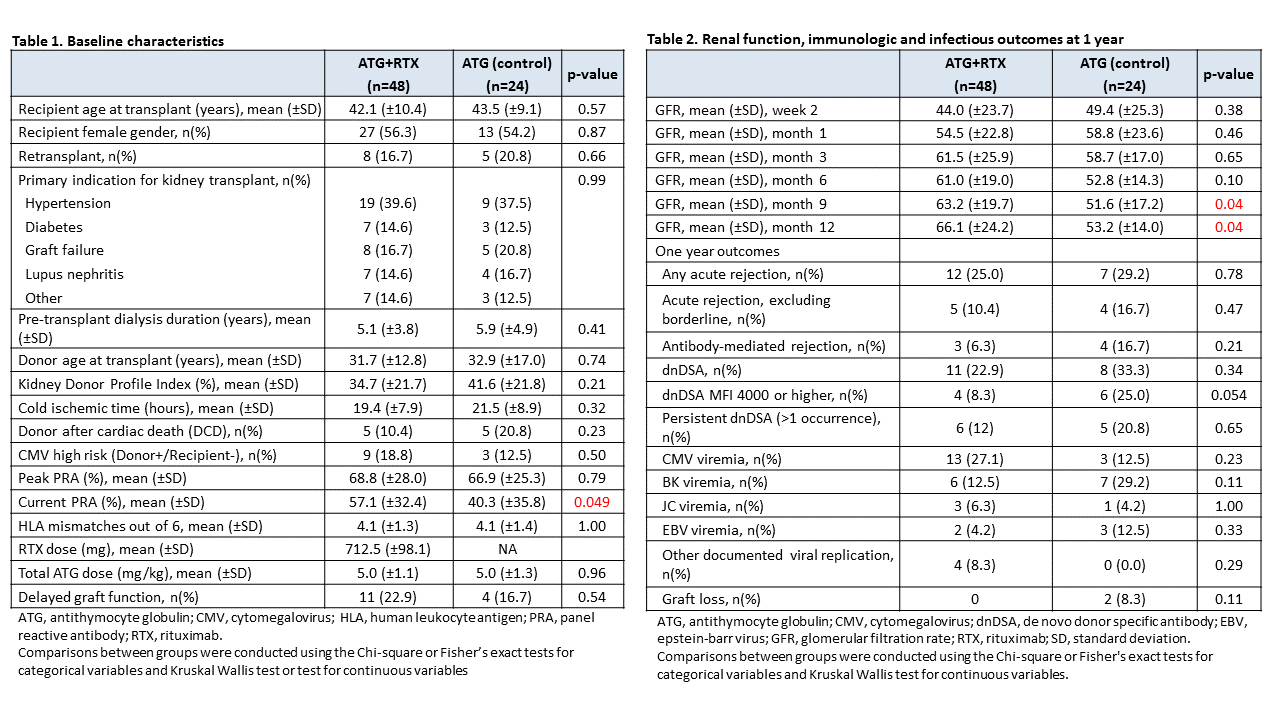
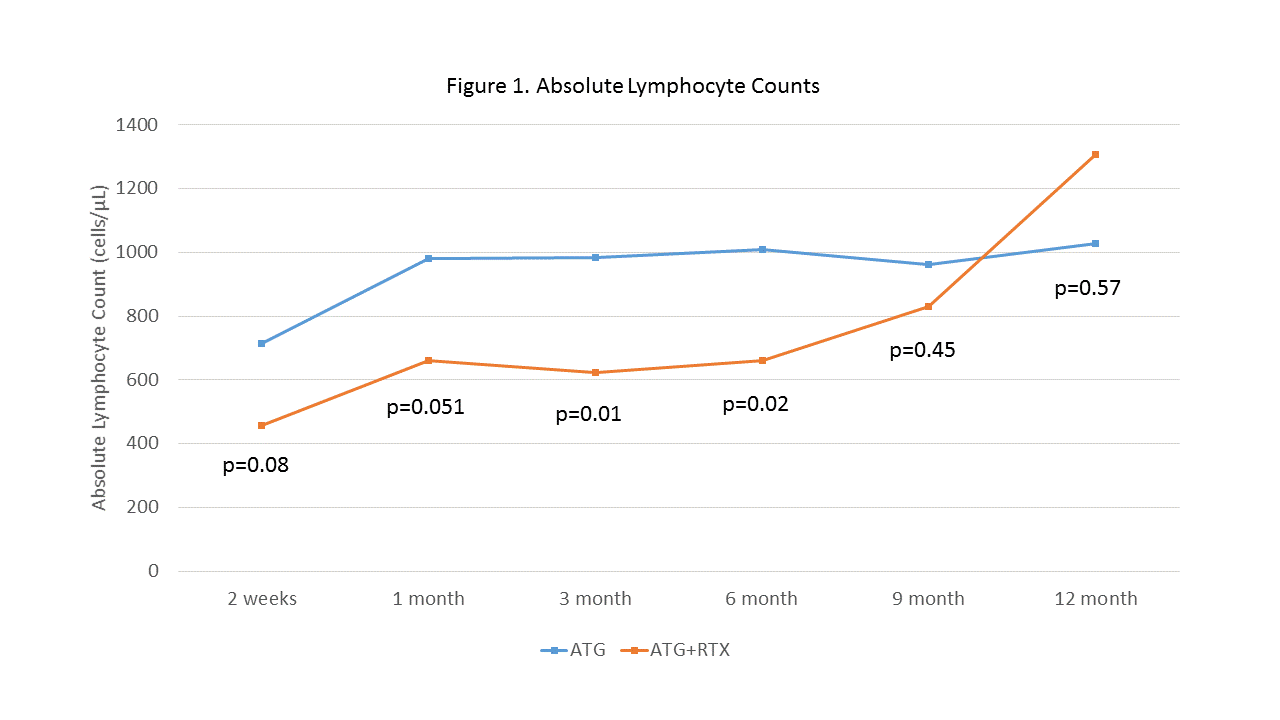
*Results: Demographics for both groups are shown in Table 1 and one year outcomes are shown in Table 2. One-year biopsy-proven rejection rates were similar between groups (ATG+RTX: 25%, ATG: 29%) while the rate of antibody mediated rejection (AMR) was numerically lower in ATG+RTX patients (6.3% vs. 16.7%, p=0.21). Similarly, dnDSA were numerically lower, particularly for dnDSA present at an MFI of 4000 or greater (8.3% vs. 25%, p=0.054). Glomerular filtration rates were significantly higher in ATG+RTX patients at 9 and 12 months and no graft losses occurred compared to 2 in the control group. A higher proportion of CMV and lower proportion of BKV were found in ATG+RTX patients; however, these differences were not statistically significant. Lymphocyte counts were lower among ATG+RTX patients for the first six months post-transplant (Figure 1).
*Conclusions: Our findings demonstrate improved renal function and possible attenuation of AMR and dnDSA in high-immunologic risk AA KTRs, though with an increased CMV risk. Continued evaluation of combined ATG+RTX induction in a larger population is ongoing.
To cite this abstract in AMA style:
Patel SJ, Kuten S, Pham C, Knight R, Hobeika M, Nguyen D, Graviss E, Yi S, Eagar T, Gaber A. Combined Antithymocyte Globulin (ATG) and Rituximab (RTX) Induction in African American (AA) Kidney Transplant Recipients [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/combined-antithymocyte-globulin-atg-and-rituximab-rtx-induction-in-african-american-aa-kidney-transplant-recipients/. Accessed May 16, 2026.« Back to 2019 American Transplant Congress