Normothermic Machine Perfusion of Genetically Modified Pig Livers with Human Blood
1Tufts Medical Center, Boston, MA, 2Massachusetts General Hospital, Boston, MA, 3eGenesis, Cambridge, MA
Meeting: 2020 American Transplant Congress
Abstract number: A-273
Keywords: knockout, Liver transplantation, Protective genes, Xenoreactive antibodies
Session Information
Session Name: Poster Session A: Xenotransplantation
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
*Purpose: Barriers to pig-to-human liver xenotransplantation remain, including profound thrombocytopenia, coagulopathy, and rejection. We utilized ex-vivo liver xenoperfusion with human blood to study the human response to genetically modified pig livers.
*Methods: Livers from wild type pigs (WT) (n=2), GTKO.hCD55 pigs (n=4) and three variations of TKO (αGal, β4Gal, CMAH) pigs with genetic modifications targeting complement, coagulation, immune, and inflammation regulation (Pig 2.0; n=6) underwent normothermic machine perfusion using freshly collected, heparinized human whole blood and plasma on the Liver Assist (Organ Assist, Groningen, NL). Perfusion was continued until graft failure, defined by decreased blood flow secondary to elevated vascular resistance. Standard laboratory analyses were performed. Tissue was collected for H&E, IgG, IgM and C4d staining.
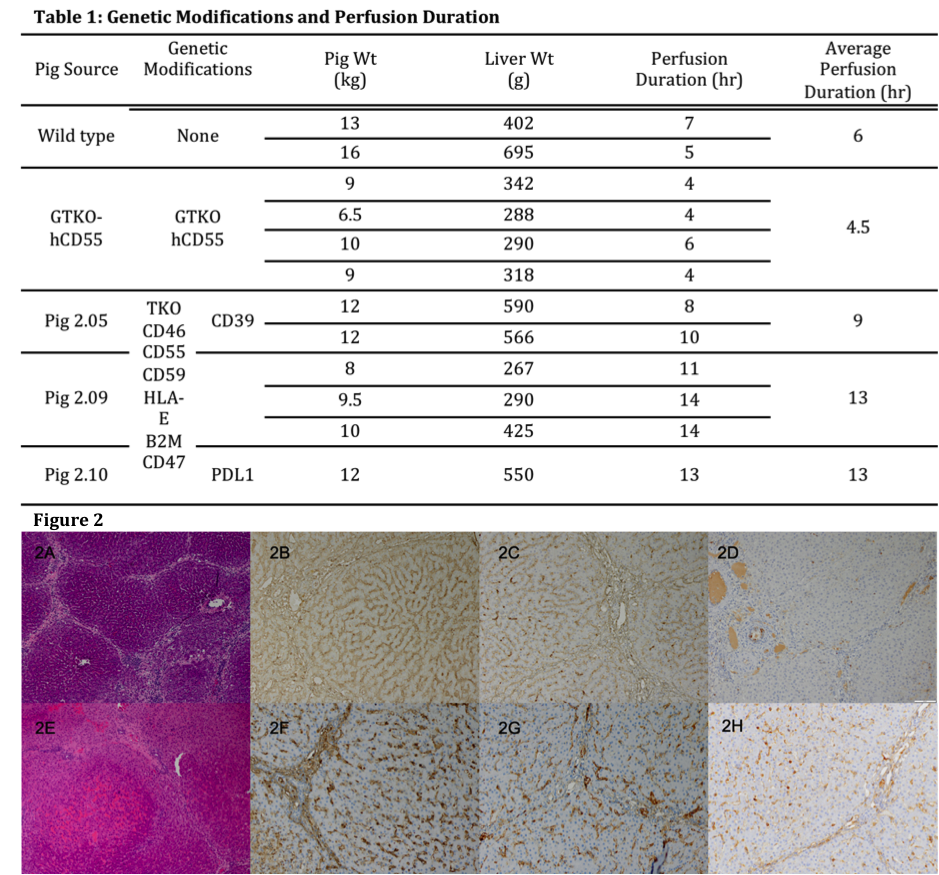
*Results: Livers in all groups demonstrated progressively decreasing blood flow. However, this was evident earlier and progressed faster in the WT and GTKO.hCD55 groups compared to the Pig 2.0 group (Table 1), which correlated to longer survival of the Pig 2.0 livers. Mean graft survival was 6 hours (range 5-7 hours) in WT livers, 4.5 hours (range 4-6 hours) in GTKO.hCD55 livers and 10 hours (range 8-14 hours) in Pig 2.0 livers. Histologically, Pig 2.0 livers exhibited preserved hepatic architecture with mild diffuse portal and sinusoidal inflammation (Fig 2A). Diffuse mild sinusoidal IgG and IgM staining confirmed antibody deposition (Fig 2B and 2C), but C4d was negative (Fig 2D). In contrast, WT livers sustained focal ischemic necrosis and vascular congestion (Fig 2E), with a greater intensity of IgM and IgG staining (Fig 2F and 2G) and C4d positivity (Fig 2H).
*Conclusions: In this ex vivo perfusion model, Pig 2.0 livers with genetic modifications targeting complement, coagulation, immune and inflammation regulation achieved prolonged function and diminished antibody and complement deposition compared to WT or GTKO.hCD55 xenografts. The prompt and profound platelet loss was not attenuated with the current modifications. This model is an efficient and informative tool to simulate pig-to-human xenotransplantation and evaluate the efficacy of different genetic constructs.
To cite this abstract in AMA style:
Detelich D, Coe TM, Rickert CG, Carroll C, Serifis N, Matheson R, Rosales I, Qin W, Kan Y, Layer J, Youd M, Westlin W, Kimura S, Azimzadeh A, Yang L, Markmann JF. Normothermic Machine Perfusion of Genetically Modified Pig Livers with Human Blood [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/normothermic-machine-perfusion-of-genetically-modified-pig-livers-with-human-blood/. Accessed May 21, 2026.« Back to 2020 American Transplant Congress