Urinary Exosomal Hemopexin, a Validated Diagnostic Marker for Rejection in Kidney Transplant Recipients
1Internal Medicine, Daegu Fatima Hospital, Daegu, Korea, Republic of, 2Internal Medicine, Kyungpook National University, Daegu, Korea, Republic of
Meeting: 2020 American Transplant Congress
Abstract number: A-312
Keywords: Biopsy, Kidney transplantation, Rejection
Session Information
Session Name: Poster Session A: Biomarkers, Immune Assessment and Clinical Outcomes
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
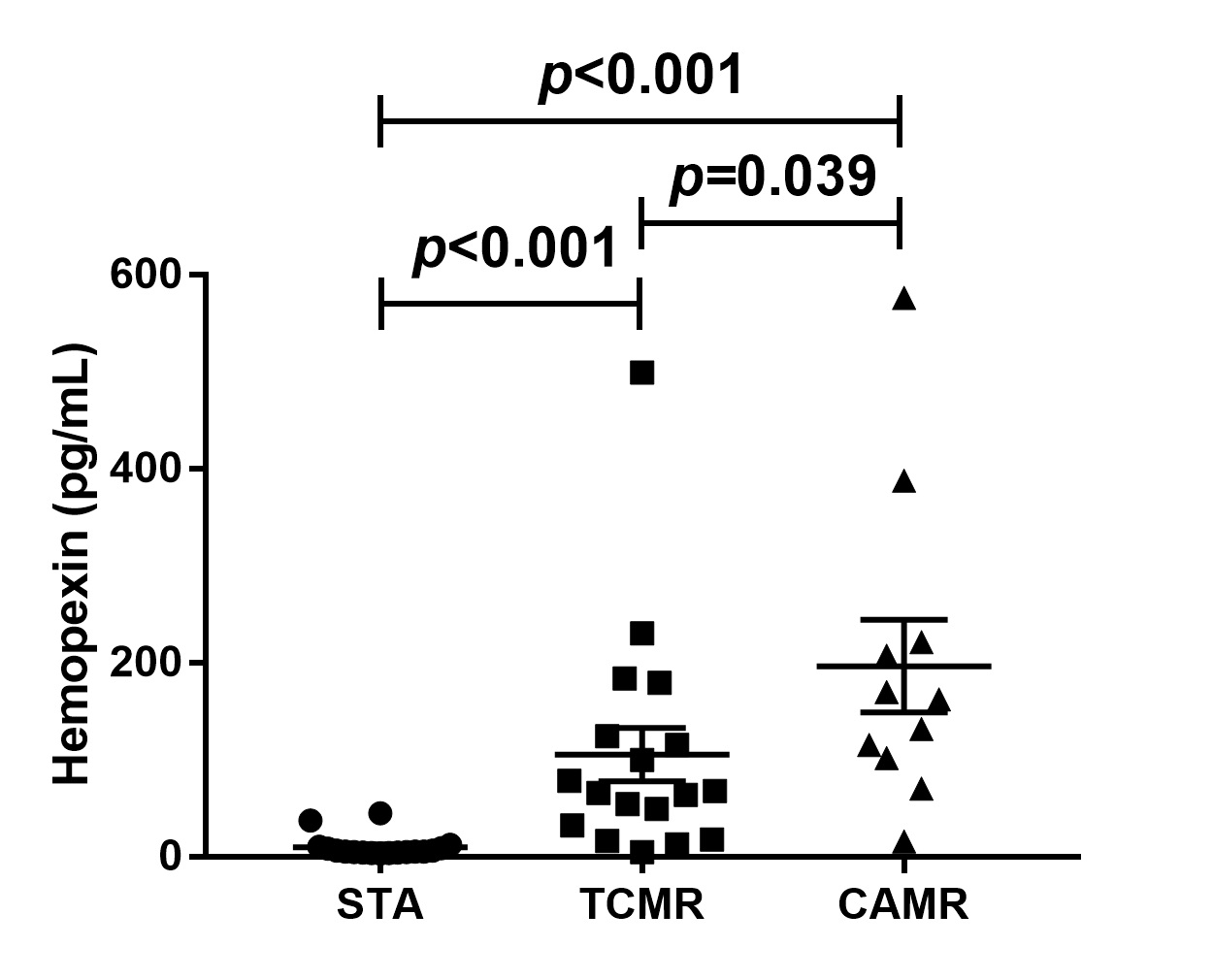
*Purpose: Urinary exosomal hemopexin was identified as a potential diagnostic candidate for kidney allograft rejection in our previous proteomic study. We validated hemopexin as a predictor of acute T cell-mediated rejection (TCMR) as well as chronic active antibody-mediated rejection (CAMR).
*Methods: Urine samples from kidney transplant recipients (KTRs) who underwent graft biopsy were analyzed. There were 98 graft biopsies and we compared the KTRs with stable graft function (STA), TCMR, and CAMR groups. Each patient’s urinary exosome was isolated by exosome isolation kit, and urinary exosomal hemopexin level was measured by ELISA method. To evaluate the discrimination ability, receiver operating characteristic (ROC) analysis was applied.
*Results: Nineteen patients were classified as STA, 18 were TCMR, and 11 were CAMR. The median urinary exosomal hemopexin level was 5.47 (interquartile, 4.30−10.22) ng/mL in STA, 66.87 (28.70−137.95) ng/mL in TCMR, and 162.07 (101.96−221.30) ng/mL in CAMR. Both TCMR and CAMR groups showed significantly higher levels of hemopexin compared to STA group (both P<0.001). In ROC analysis, urinary exosomal hemopexin in TCMR showed an area under curve of 0.90 (95% CI 0.78−1.00) and 0.95 (0.86−1.00) in CAMR.
*Conclusions: Urinary exosomal hemopexin levels were increased in TCMR and CAMR. It could be a potential diagnostic marker for both TCMR and CAMR.
To cite this abstract in AMA style:
Lee J, Kim J, Lee G, Jeon S, Noh H, Lim J, Jung H, Choi J, Cho J, Park S, Kim Y, Kim C. Urinary Exosomal Hemopexin, a Validated Diagnostic Marker for Rejection in Kidney Transplant Recipients [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/urinary-exosomal-hemopexin-a-validated-diagnostic-marker-for-rejection-in-kidney-transplant-recipients/. Accessed May 26, 2026.« Back to 2020 American Transplant Congress