The Role of Donor-Derived Cell-Free DNA in Identification of Injury in Kidney Allografts with Antibody-Mediated Rejection and De Novo DSA
1Organ Transplant Center, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China
2BGI-Guangzhou Medical Laboratory, BGI-Shenzhen, Guangzhou, China.
Meeting: 2018 American Transplant Congress
Abstract number: 22
Keywords: Antibodies, Kidney transplantation, Rejection, Renal injury
Session Information
Session Name: Concurrent Session: Kidney Acute Antibody Mediated Rejection
Session Type: Concurrent Session
Date: Sunday, June 3, 2018
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:06pm-3:18pm
Presentation Time: 3:06pm-3:18pm
Location: Room Hall 4B
Objectives: To evaluate the capacity of donor-derived cell-free DNA (dd-cfDNA) in the identification of injury in kidney allografts with antibody-mediated rejection and de novo DSA.
Methods: Blood specimens were collected in kidney allograft recipients with either antibody-mediated rejection (AMR) or de novo DSA (dnDSA) without histological lesions. The plasma level of dd-cfDNA in recipients was measured using a panel of SNP genotyping we developed, independent of the donor blood specimens.
Results: Thirty-seven recipients were enrolled for analysis (18 AMR, 7 dnDSA and 12 controls). A significant increased level of dd-cfDNA was identified in AMR group or dnDSA group compared with control group 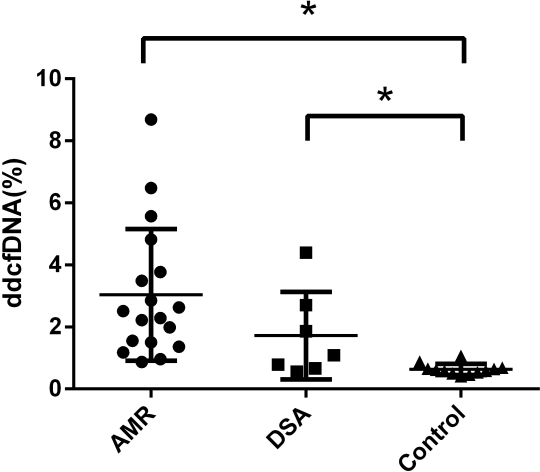 (AMR 3.04%±2.12% vs Control 0.63%±0.18%, P<0.05; AMR 1.72%±1.41% vs Control 0.63%±0.18%, P<0.05; ANOVA P<0.01). The receiver operating characteristic area under the curve (AUC) was 0.97 (95% confidence interval [95%CI], 0.96 to 1.00) and 0.84 (95%CI, 0.65 to 1.00) respectively for the discrimination of AMR or dnDSA from the control group. Positive and negative predictive values for AMR at a cutoff of 1.0% dd-cfDNA were 94.1% and 84.6%, respectively. Positive and negative likelihood ratios for AMR at a cutoff of 1.0% dd-cfDNA were 10.7 and 0.12, respectively.
(AMR 3.04%±2.12% vs Control 0.63%±0.18%, P<0.05; AMR 1.72%±1.41% vs Control 0.63%±0.18%, P<0.05; ANOVA P<0.01). The receiver operating characteristic area under the curve (AUC) was 0.97 (95% confidence interval [95%CI], 0.96 to 1.00) and 0.84 (95%CI, 0.65 to 1.00) respectively for the discrimination of AMR or dnDSA from the control group. Positive and negative predictive values for AMR at a cutoff of 1.0% dd-cfDNA were 94.1% and 84.6%, respectively. Positive and negative likelihood ratios for AMR at a cutoff of 1.0% dd-cfDNA were 10.7 and 0.12, respectively.
Conclusions: Donor-derived cfDNA may be used to assess allograft injury in AMR and recipients with dnDSA but no histological lesions. Donor-derived cfDNA levels <1% reflect the absence of AMR and levels >1% indicate a probability of injury in dnDSA (+) recipients.
Figure legend:
Figure 1. The plasma level of donor-derived cell-free DNA (dd-cfDNA) in antibody-mediated rejection (AMR), de novo DSA and control group. *Significant at a level of 0.05.
CITATION INFORMATION: Zhang H., Liu L., Zheng C., Li X., Fu Q., Li J., Su Q., Huang H., Ye M., Wang C. The Role of Donor-Derived Cell-Free DNA in Identification of Injury in Kidney Allografts with Antibody-Mediated Rejection and De Novo DSA Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Zhang H, Liu L, Zheng C, Li X, Fu Q, Li J, Su Q, Huang H, Ye M, Wang C. The Role of Donor-Derived Cell-Free DNA in Identification of Injury in Kidney Allografts with Antibody-Mediated Rejection and De Novo DSA [abstract]. https://atcmeetingabstracts.com/abstract/the-role-of-donor-derived-cell-free-dna-in-identification-of-injury-in-kidney-allografts-with-antibody-mediated-rejection-and-de-novo-dsa/. Accessed May 15, 2026.« Back to 2018 American Transplant Congress
