Prognostic Value of Dd-cfDNA Assay in Acute Renal Rejection Therapy: A Prospective Cohort Study
Kidney Disease Center, First Affiliated Hospital of Zhejiang University, Hangzhou, China
Meeting: 2020 American Transplant Congress
Abstract number: A-346
Keywords: Kidney transplantation, Tolerance
Session Information
Session Name: Poster Session A: Acute Rejection
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
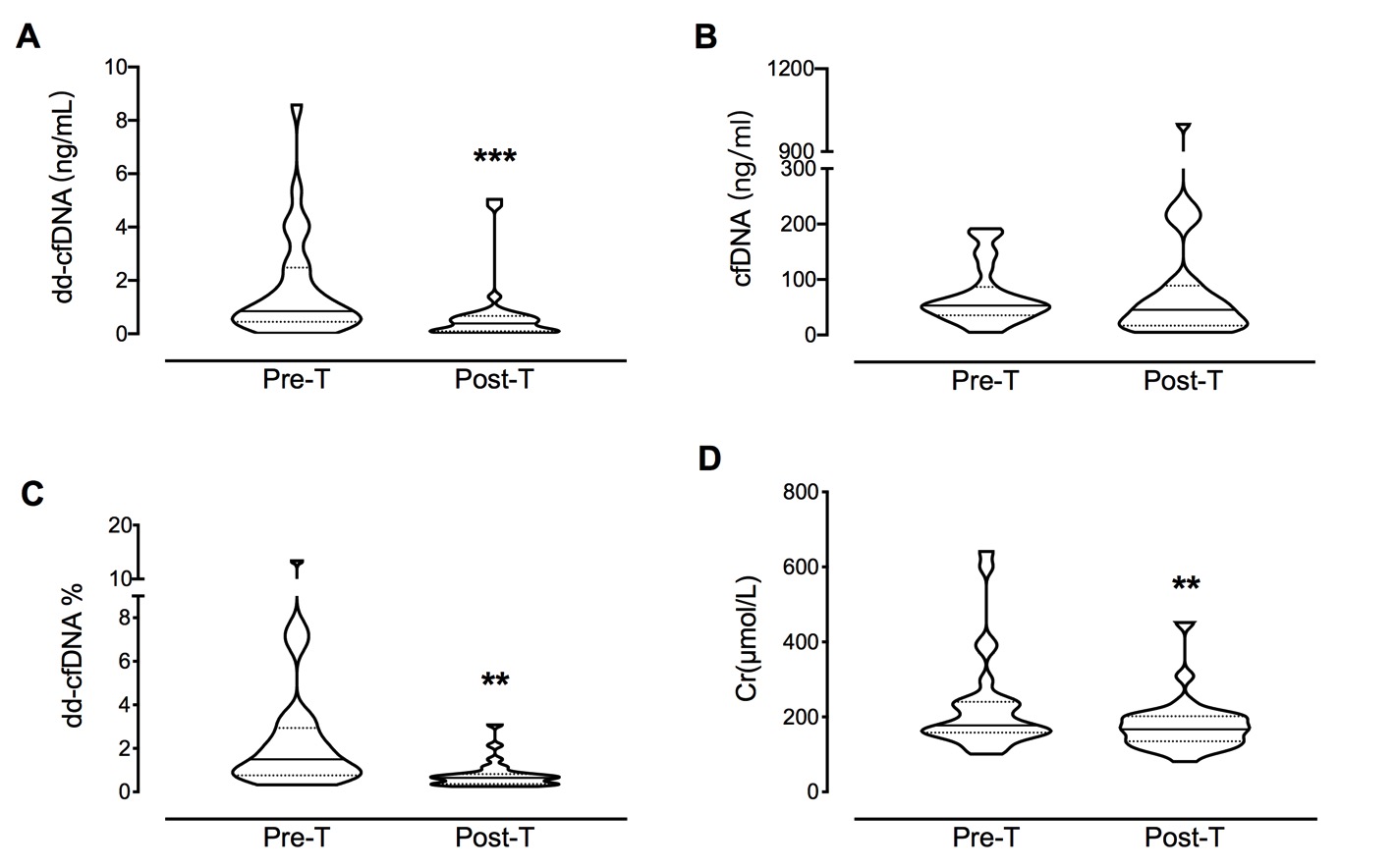
*Purpose: Donor-derived cell free DNA (dd-cfDNA) is a promising biomarker for monitoring allograft status. However, whether it is capable of reflecting real time anti-rejection treatment effects still remains unclear.
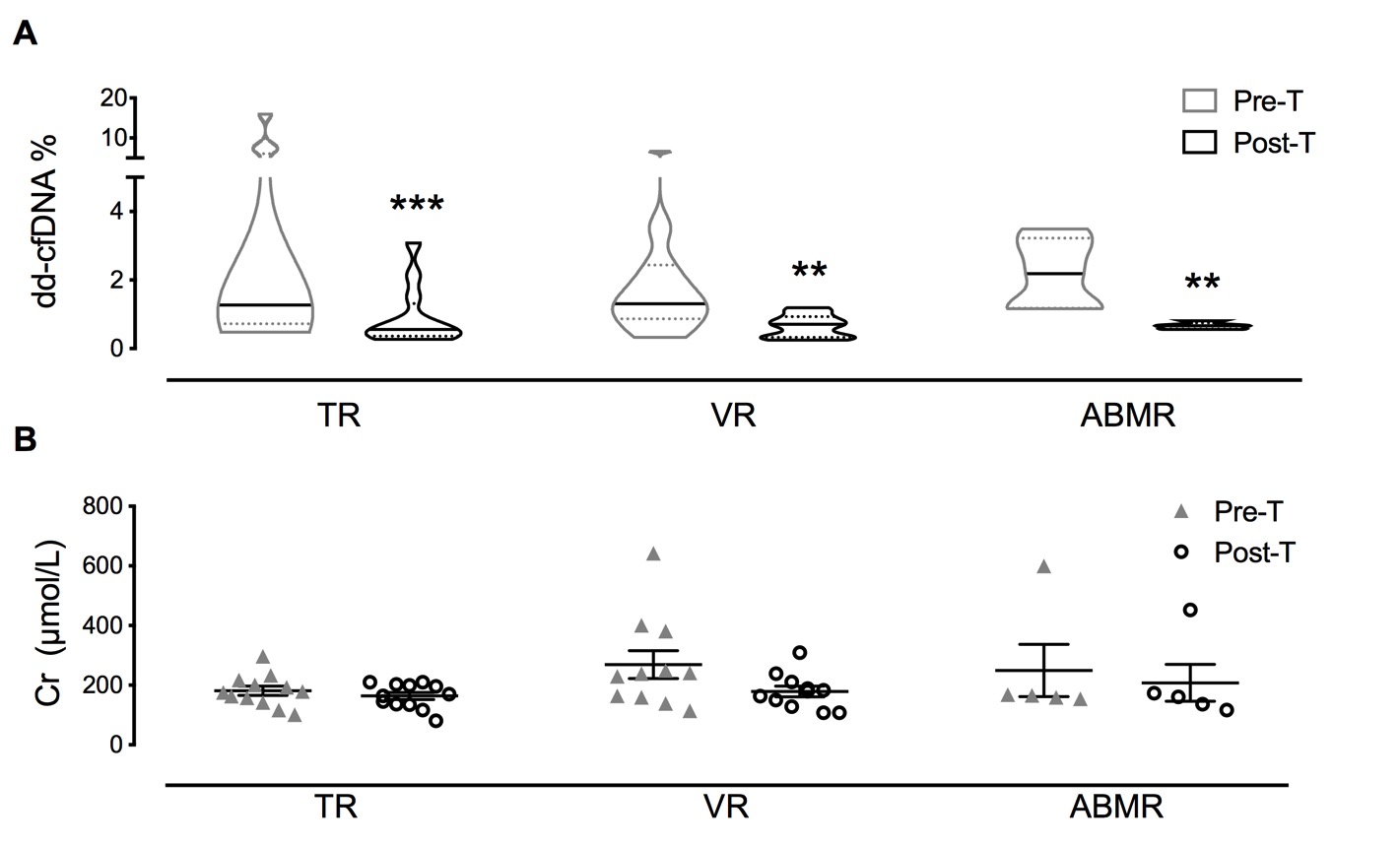
*Methods: We prospectively included 28 patients with acute renal rejection, of which included 5 with ABMR, 12 with type IA or type IB rejection (TR), and 11 with type IIA or IIB rejection (VR). The dd-cfDNA levels in peripheral blood were measured by human SNP loci capture hybridization.
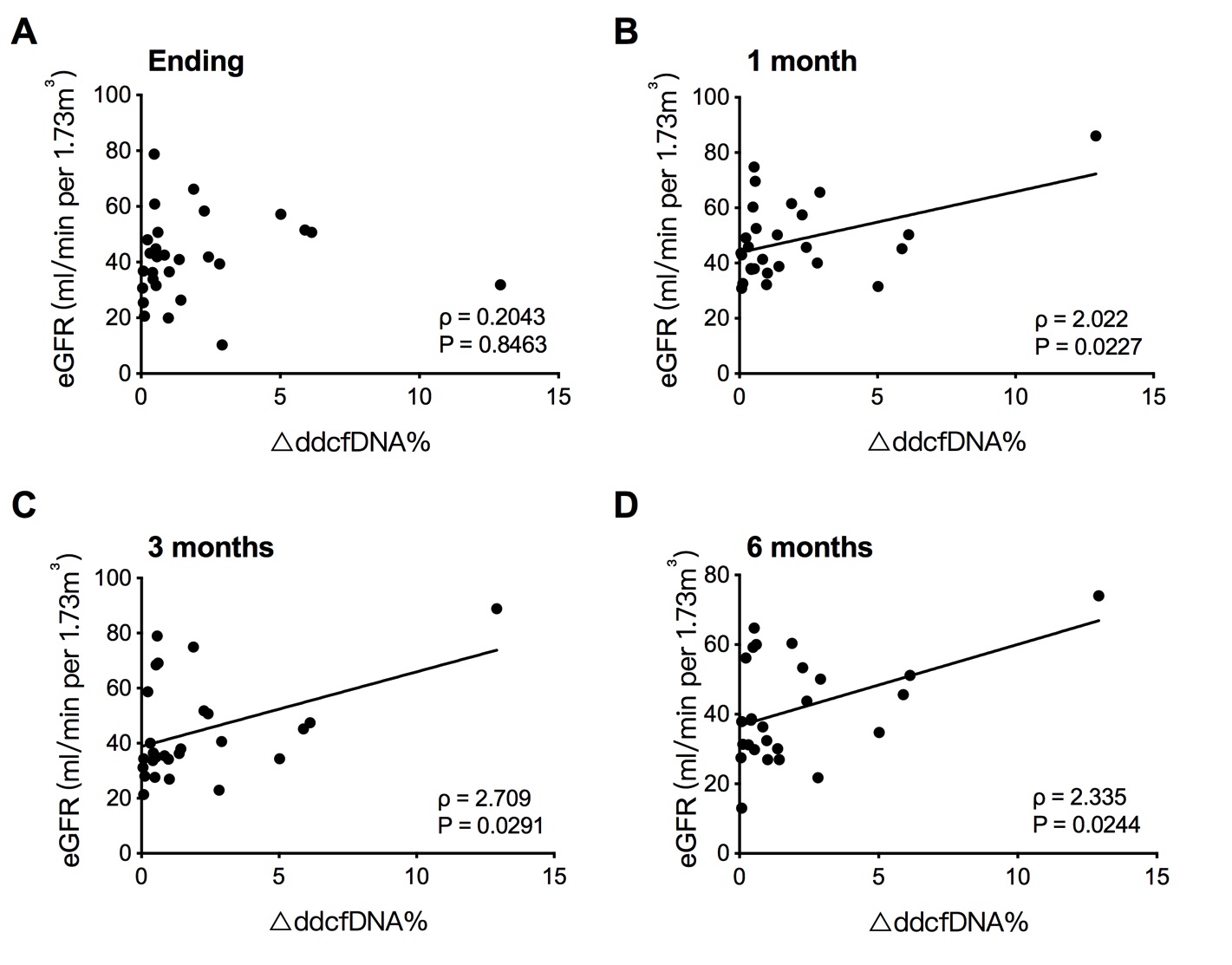
*Results: The percentage of dd-cfDNA (dd-cfDNA%) declined significantly from 2.566 ± 0.549% to 0.773 ± 0.116% (P < 0.001) after anti-rejection therapy. The dd-cfDNA% reduced steadily over the course of 3 days with daily methylprednisolone injections, while it showed no significant difference between the end of anti-rejection therapy and 2 weeks later. Furthermore, the changes of dd-cfDNA% (∆dd-cfDNA%) demonstrated a positive correlation with eGFR at 1 month (ρ =2.570, P = 0.022), 3 months (ρ = 3.210, P = 0.027), and 6 months (ρ = 2.860, P = 0.019) after therapy.
*Conclusions: The dd-cfDNA assay showed the prognostic capabilities in therapy outcome and allograft recovery, but its ability is inhibited by methylprednisolone regardless of the types of rejection and reassessment of frequency intervals needs to be conducted.
To cite this abstract in AMA style:
Guo L, Wang R, Lv J, Shen J, Wu J, Chen J. Prognostic Value of Dd-cfDNA Assay in Acute Renal Rejection Therapy: A Prospective Cohort Study [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/prognostic-value-of-dd-cfdna-assay-in-acute-renal-rejection-therapy-a-prospective-cohort-study/. Accessed May 17, 2026.« Back to 2020 American Transplant Congress