Novel Treatment of Acute Antibody-Mediated Rejection in a Nonhuman Primate Model of Kidney Allotransplantation
1Department of Surgery, Duke University Medical Center, Durham, NC, 2Department of Pathology and Laboratory Medicine, University of Pennsylvania, Philadelphia, PA
Meeting: 2020 American Transplant Congress
Abstract number: 25
Keywords: Alloantibodies, B cells, Co-stimulation, T helper cells
Session Information
Session Name: Kidney Acute Antibody Mediated Rejection
Session Type: Oral Abstract Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:45pm
 Presentation Time: 4:27pm-4:39pm
Presentation Time: 4:27pm-4:39pm
Location: Virtual
*Purpose: One third of sensitized, HLA-incompatible transplant recipients experience acute antibody mediated rejection (AMR) with limited treatment options. We tested a novel treatment strategy for AMR consisting of complement inhibition, proteasome inhibition and costimulation blockade in a nonhuman primate model.
*Methods: Three (3) rhesus macaques were sensitized to maximally MHC mismatched donors by two sequential skin transplants. Primates subsequently received kidney allografts from their skin donors without prior desensitization. All primates were maintained on tacrolimus, MMF and steroids immunosuppression. Primates were monitored postoperatively for signs of acute AMR, which was defined as worsening kidney function resistant to high dose steroid rescue therapy and a rise in serum donor-specific antibody (DSA) levels. Our AMR treatment consisted of a combination of the C3 complement inhibitor Compstatin for 1 week together with carfilzomib and belatacept for 4 weeks.
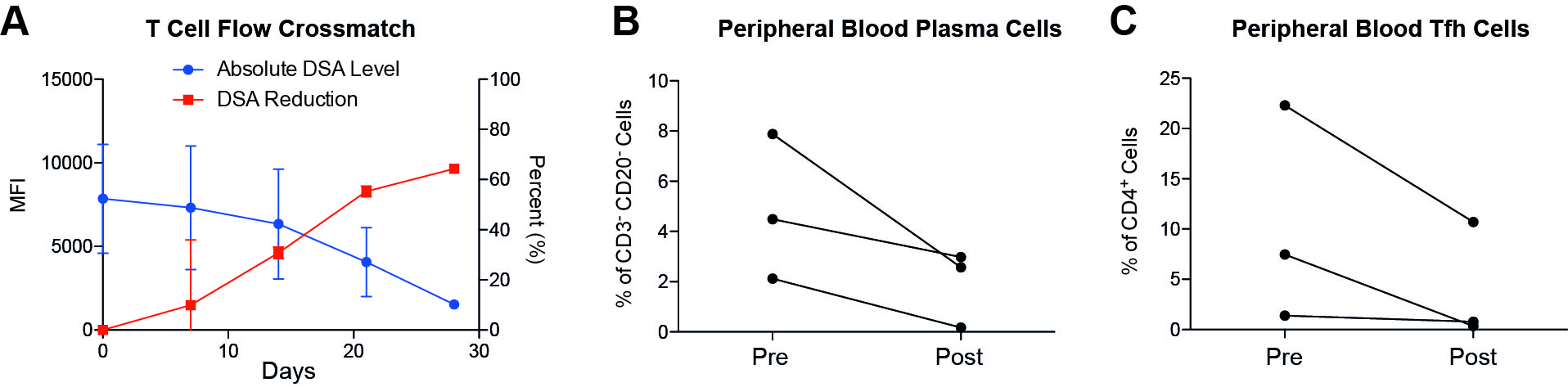
*Results: The included primates began treatment on days 19, 40 and 48, respectively. The combination of Compstatin, carfilzomib and belatacept was tolerated well and we did not observe any treatment-related toxicity. After initiation of treatment, we observed a reduction of class I and class II DSA in all primates (Figure 1A). Additionally, we observed reduction of peripheral blood plasma cells, defined as CD20–CD19+CD38+ cells, by flow cytometry (Figure 1B), as well as peripheral blood T follicular helper cells (Figure 1C). Most importantly, primates experienced improvement of kidney function as evidenced by reduced serum creatinine and BUN as well as increased urine output. Our treatment extended the graft survival by up to 2 months.
*Conclusions: The combination of Compstatin, carfilzomib and belatacept was able to control the humoral immune response seen during acute AMR in a sensitized nonhuman primate model. This was associated with improved graft function and prolongation of graft survival.
To cite this abstract in AMA style:
Schmitz R, Fitch ZW, Choi AY, Manook M, Yoon J, Lambris JD, Kwun J, Knechtle SJ. Novel Treatment of Acute Antibody-Mediated Rejection in a Nonhuman Primate Model of Kidney Allotransplantation [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/novel-treatment-of-acute-antibody-mediated-rejection-in-a-nonhuman-primate-model-of-kidney-allotransplantation/. Accessed March 25, 2026.« Back to 2020 American Transplant Congress