Impact of Anti-CMV Prophylactic Therapy on the Development of CMV Quantiferon Sero-Conversion in High-Risk Patients
Transplant Surgery, Medical University of South Carolina, Charleston, SC.
Meeting: 2015 American Transplant Congress
Abstract number: A289
Keywords: Cytomeglovirus, Infection
Session Information
Session Name: Poster Session A: Late Breaking
Session Type: Poster Session
Date: Saturday, May 2, 2015
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Exhibit Hall E
Body: Abdominal organ transplants at high risk for CMV infection (D+/R) remain a difficult cohort to prevent late CMV infection despite prolonged valganciclovir (VGC) administration. Administration of passive immunity using CMV hyperimmune globulin (CMVIgG) may provide adequate prophylaxis and allow for development of active immunity in these at risk patients. The aim of this study was to evaluate the cumulative incidence of sero-conversion of quantiferon in D+/R abdominal transplant recipients administered VCG for 100 days posttransplant in combination with CMVIgG versus VCG for 200 days.
Methods: Prospective, randomized, open label pilot study of 40 consecutively enrolled adult abdominal transplant recipients. Patients were randomized and stratified based on organ to VCG per package insert guidelines for 200 days vs VCG for 100 days posttransplant with CMVIgG 100 mg/kg administered at 90 days, 120 days, and 180 days posttransplant. Whole blood interferon-gamma release assay using QuantiFERON-CMV was performed at serial time points to evaluate the cumulative incidence of sero-conversion.
Results: Of the 40 patients enrolled, a total of 6 patients developed CMV viremia. Viremia occurred in 2 patients in the CMV IgG group after 200 days (216 and 260 days posttransplant). There were similar rates of opportunistic infections and hospitializations in the kidney recipients between groups, but numerically more in the liver CMV IgG group (Table 2). CMV syndrome and disease before and after 200 days were modest in all groups (Table 2). Compared to VGC treatment, cumulative incidence of quantiferon sero-conversion was significantly higher in CMVIgG group at 180 days post-transplant (Figure1).
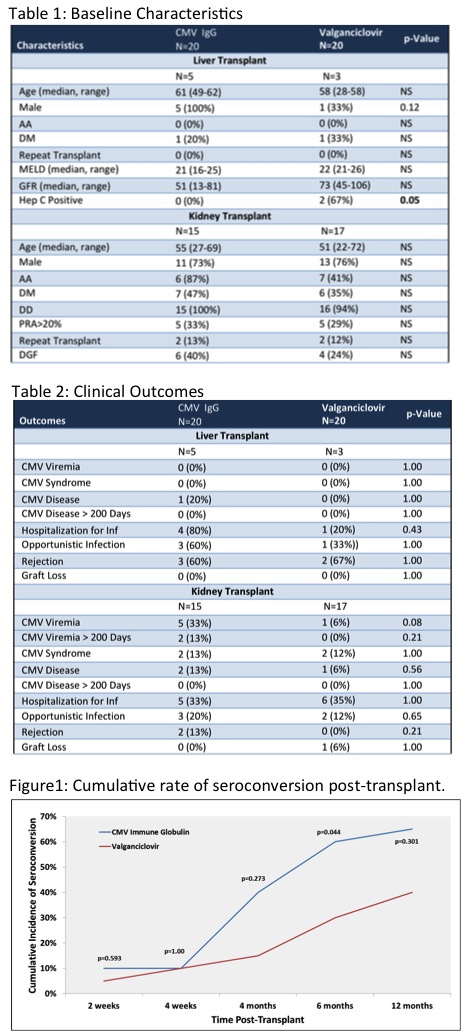
Conclusion: CMV IgG may provide a better alternative to VCG in high-risk abdominal organ transplant recipients, due to equal efficacy rates and earlier CMV quantiferon sero-conversion. Larger trials are needed to confirm these results.
To cite this abstract in AMA style:
Palanisamy A, Taber D, Pilch N, Bratton C, McGillicuddy J, Baliga P, Chavin K. Impact of Anti-CMV Prophylactic Therapy on the Development of CMV Quantiferon Sero-Conversion in High-Risk Patients [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/impact-of-anti-cmv-prophylactic-therapy-on-the-development-of-cmv-quantiferon-sero-conversion-in-high-risk-patients/. Accessed May 19, 2026.« Back to 2015 American Transplant Congress
