Evaluation of Leflunomide for the Treatment of BK Viremia and Biopsy Proven BK Nephropathy: A Single Center Experience
1Saint Louis University Hospital, St. Louis, MO
2Minneapolis VA Health Care System, Minneapolis, MN.
Meeting: 2015 American Transplant Congress
Abstract number: A31
Keywords: Polyma virus
Session Information
Session Name: Poster Session A: BK Virus Infection
Session Type: Poster Session
Date: Saturday, May 2, 2015
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Exhibit Hall E
Purpose:
The objective was to evaluate the use of leflunomide in BK viremia (BKV) and biopsy proven BK nephropathy (BKN) in kidney and kidney-pancreas transplant recipients.
Methods:
We retrospectively reviewed 28 kidney and kidney-pancreas transplant recipients that received leflunomide for BKV from January 2006 to November 2012. Demographics, time to BKV diagnosis, biopsy history, rejection episodes, and laboratory data were recorded.
Results:
The average (mean ± SD) time to BKV diagnosis from time of transplant was 316.1 ± 368.0 days (62-1708). At time of diagnosis, 64% of patients had their maintenance immunosuppression reduced. Leflunomide was started in patients with BKV and biopsy proven acute rejection (BPAR) (50%) or biopsy proven BKN (18%). Therapeutic level (50-100 mcg/mL) was achieved in 54% of patients, and 60% of these required a leflunomide dose of at least 60 mg/day. BK virus was cleared from the serum on average of 151 ± 145.2 days (17-476). Twenty-nine percent of patients never cleared the virus. At study commencement, 29% of patients remained on leflunomide due to persistent BKV or as maintenance immunosuppression.
Conclusion:
In our study, most patients required at least a 60 mg daily dose of leflunomide to achieve therapeutic levels and to clear the virus compared to the standard 40 mg daily dose. Delaying therapy may result in progressive BKV or BKN. 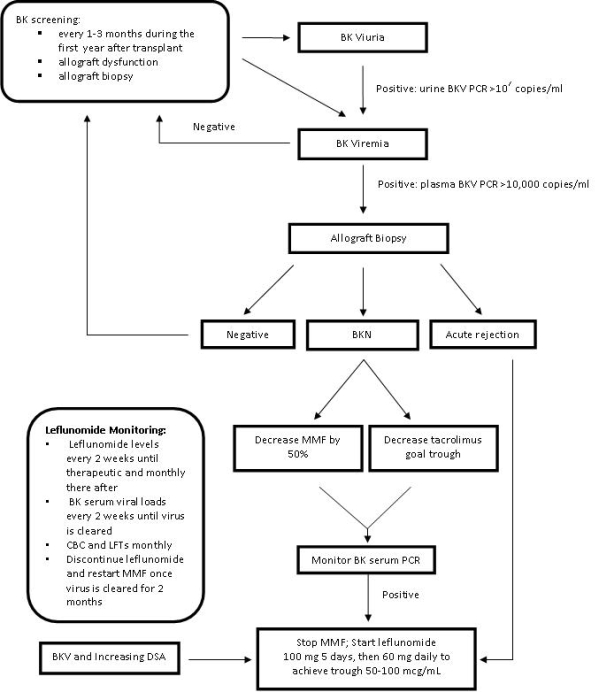
To cite this abstract in AMA style:
Nesselhauf N, Strutt J, Bastani B. Evaluation of Leflunomide for the Treatment of BK Viremia and Biopsy Proven BK Nephropathy: A Single Center Experience [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/evaluation-of-leflunomide-for-the-treatment-of-bk-viremia-and-biopsy-proven-bk-nephropathy-a-single-center-experience/. Accessed May 21, 2026.« Back to 2015 American Transplant Congress
