Evaluating Outcomes in Multi-Organ Liver Transplant Recipients
1SRTR, MMRF, Minneapolis, MN
2University of MN, Minneapolis, MN
3Stanford School of Medicine, Stanford, CA.
Meeting: 2015 American Transplant Congress
Abstract number: A237
Keywords: Liver transplantation, Survival
Session Information
Session Name: Poster Session A: Non Organ Specific, Economics, Public Policy, Allocation, Ethics
Session Type: Poster Session
Date: Saturday, May 2, 2015
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Exhibit Hall E
Historically, transplant (Tx) program evaluations performed by the Scientific Registry of Transplant Recipients (SRTR) in the US have included only single-organ Txs, except for simultaneous pancreas-kidney (SPK) and heart-lung Txs. Recognizing the absence of publicly available data on multi-organ (MO) Txs, the SRTR Technical Advisory Committee recommended that SRTR explore ways to evaluate MO Txs. Because simultaneous liver-kidney (SLK) Txs are the second-most common after SPK, we began by considering ways to evaluate MO liver Tx outcomes. We evaluated program performance by 1) including MO liver Txs with the single-organ Txs, and 2) evaluating performance on MO Txs independently from single-organ Txs. All deceased-donor liver Txs performed 1/1/2011-6/30/2013 were included (N=13930); 8% were SLK (N=1084) and 1% were other types of MO Txs (N=98). For single-organ liver Txs, expected outcomes were estimated using the standard SRTR risk adjustment models. For MO recipients, the standard program-specific report model was refit on the MO cohort with additional adjusters for whether the Tx was an SLK or another type of MO Tx. To compare outcomes, Bayesian hazard ratios (HR) were estimated using the newly adopted SRTR methodology, and programs were hypothetically identified for review if they met the newly adopted Membership and Professional Standards Committee methodology (P[HR>1.2]>0.75 or P[HR>2.5]>0.1). Of 112 liver programs, 14 (12%) had 1-year graft failure rates high enough to meet the flagging threshold for their single-organ Txs, and 8 programs met the threshold for their MO Txs. Correlation between single-organ and MO HRs was low (0.192, p=0.05, Figure). 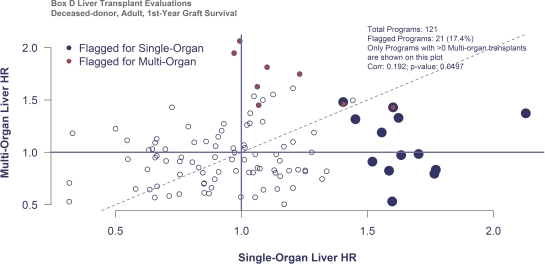 Alternatively, if MO Txs are combined with single-organ Txs, 11 programs would meet review criteria, missing 9 with apparent underperformance on single-organ or MO Txs. Given the low correlation between evaluations on single-organ and MO recipients, reporting outcomes separately in these subpopulations is preferred over combining MO Txs with the single-organ Txs.
Alternatively, if MO Txs are combined with single-organ Txs, 11 programs would meet review criteria, missing 9 with apparent underperformance on single-organ or MO Txs. Given the low correlation between evaluations on single-organ and MO recipients, reporting outcomes separately in these subpopulations is preferred over combining MO Txs with the single-organ Txs.
To cite this abstract in AMA style:
Snyder J, Salkowski N, Zaun D, Thompson B, Zeglin J, Schladt D, Lake J, Kim W, Israni A, Kasiske B. Evaluating Outcomes in Multi-Organ Liver Transplant Recipients [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/evaluating-outcomes-in-multi-organ-liver-transplant-recipients/. Accessed May 18, 2026.« Back to 2015 American Transplant Congress
