Early Conversion to Everolimus in De Novo Renal Transplant Recipients: Incidence of Viral Infections in the Randomized-Controlled ELEVATE Study.
1ELEVATE Study Group, Basel, Switzerland
2Novartis, Basel, Switzerland
3Novartis Pharmaceutical Corporation, East Hanover.
Meeting: 2016 American Transplant Congress
Abstract number: 257
Keywords: Calcineurin, Immunosuppression, Infection, Kidney transplantation
Session Information
Session Name: Concurrent Session: Various Viruses, Vaccines, and SOT
Session Type: Concurrent Session
Date: Monday, June 13, 2016
Session Time: 2:30pm-4:00pm
 Presentation Time: 2:54pm-3:06pm
Presentation Time: 2:54pm-3:06pm
Location: Room 306
Background
Cytomegalovirus (CMV) and BK polyomavirus (BKPyV) infections are independent risk factors for acute rejection and graft loss. Early conversion from calcineurin inhibitors (CNIs) to everolimus (EVR) may protect the kidney allograft from CNI-related damage and reduce the risk of viral infections. Here we evaluate the incidence of viral infections at 24-months (M) post-transplantation (Tx), from the ELEVATE study.
Methods
ELEVATE (NCT01114529) was a 24M, open-label, multicenter study, in which de novo kidney Tx recipients (KTxR) were randomized at 10-14 weeks post-Tx to receive either EVR (n=360; trough level [C0] 6-10 ng/mL) or continue standard CNI regimen (n=357; C0: TAC 5-10 ng/mL or CsA 100-250 ng/mL). A key safety assessment was the incidence, severity, and quantification of CMV, BKPyV, and Epstein-Barr virus (EBV) infections.
Results
At M24, the incidence of overall viral infections was lower in the EVR arm (18.8 vs. 22.6%). CMV (4.3 vs. 7.8%) and BKPyV infections (1.4 vs. 3.3%) were lower in the EVR arm; no EBV infections were recorded in either arm.
There were no 'severe' CMV (0% vs. 1.1%) and BKPyV infections (0 vs. 0.3%) in the EVR vs. CNI arm. There were no CMV infections (0 vs. 0.6%) and BKPyV infections (0 vs. 0.8%) that led to the study drug discontinuation in the EVR arm compared with the CNI arm.
The median CMV viral load was lower in the EVR arm. The median BKPyV viral load was lower in the EVR arm compared with CNI arm in viremic and significantly lower in the viruric patients (P=0.0016). The median EBV viral load was higher in the EVR arm.
Conclusions
At 24M post-Tx, the incidence of overall viral (including CMV and BKPyV) infections was lower in the EVR arm. These findings are consistent with what was previously observed in kidney transplant studies involving EVR. Early conversion from CNI to EVR was associated with lower CMV and BKPyV loads. 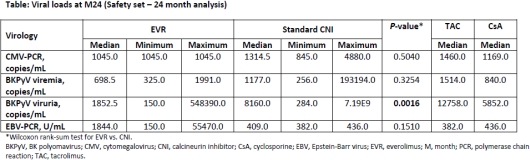
CITATION INFORMATION: Hirsch H, De Fijter J, van der Giet M, Zeier M, Pascual J, Avihingsanon Y, Lopez P, Aguilar-Sanchez J, Kochuparampil J, Wang Z, Holdaas H, Cruzado J. Early Conversion to Everolimus in De Novo Renal Transplant Recipients: Incidence of Viral Infections in the Randomized-Controlled ELEVATE Study. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Hirsch H, Fijter JDe, Giet Mvander, Zeier M, Pascual J, Avihingsanon Y, Lopez P, Aguilar-Sanchez J, Kochuparampil J, Wang Z, Holdaas H, Cruzado J. Early Conversion to Everolimus in De Novo Renal Transplant Recipients: Incidence of Viral Infections in the Randomized-Controlled ELEVATE Study. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/early-conversion-to-everolimus-in-de-novo-renal-transplant-recipients-incidence-of-viral-infections-in-the-randomized-controlled-elevate-study/. Accessed June 2, 2026.« Back to 2016 American Transplant Congress
