Discrepancies Between the U.S. Transplant Registry and Cancer Registries Regarding Cancer Diagnoses in Transplant Recipients
1National Cancer Institute, Bethesda
2Johns Hopkins Hospital, Baltimore
3SRTR, Minneapolis.
Meeting: 2015 American Transplant Congress
Abstract number: D224
Keywords: Malignancy, Risk factors
Session Information
Session Name: Poster Session D: PTLD/Malignancies: All Topics
Session Type: Poster Session
Date: Tuesday, May 5, 2015
Session Time: 5:30pm-6:30pm
 Presentation Time: 5:30pm-6:30pm
Presentation Time: 5:30pm-6:30pm
Location: Exhibit Hall E
Background: Transplant recipients have elevated cancer risk. US transplant centers report incident cancer diagnoses to the transplant network at yearly intervals, and these data are collected in the Scientific Registry of Transplant Recipients (SRTR). SRTR cancer data are used in research, but their completeness and accuracy are unknown.
Methods: The Transplant Cancer Match Study links the SRTR to 15 US cancer registries (N=208,994 transplants, followed during 1987-2010). We evaluated 8 common cancers (non-Hodgkin lymphoma [NHL], melanoma, and cancers of the colorectum, liver, lung, breast, prostate, and kidney) that were diagnosed in recipients following transplantation, as documented by the SRTR and/or cancer registries. Kappa statistics were used to assess agreement.
Results: As documented in either the SRTR or cancer registries, the number of cases ranged from 322 for liver cancer to 2283 for NHL. 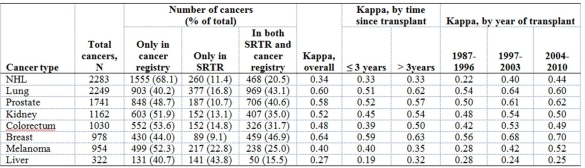 Across the various cancers, only 16-47% of cases were documented in both the SRTR and cancer registries. The proportion of cases only in the cancer registries was consistently high (40-68%) and larger than the proportion only in the SRTR (9-44%). Kappa ranged from 0.27 for liver cancer and 0.34 for NHL, to 0.60 for lung cancer and 0.64 for breast cancer. Kappa for NHL improved to 0.50 when 821 diagnoses of post-transplant lymphoproliferative disorder in the SRTR were added. For most cancers, kappa did not differ by time since transplant, but kappa improved in more recent calendar years (Table).
Across the various cancers, only 16-47% of cases were documented in both the SRTR and cancer registries. The proportion of cases only in the cancer registries was consistently high (40-68%) and larger than the proportion only in the SRTR (9-44%). Kappa ranged from 0.27 for liver cancer and 0.34 for NHL, to 0.60 for lung cancer and 0.64 for breast cancer. Kappa for NHL improved to 0.50 when 821 diagnoses of post-transplant lymphoproliferative disorder in the SRTR were added. For most cancers, kappa did not differ by time since transplant, but kappa improved in more recent calendar years (Table).
Conclusion: Based on a comparison to cancer registries, the US transplant registry is missing a large number of cancers in transplant recipients, and agreement between these two data sources regarding cancer diagnoses is often only fair. Although ascertainment of post-transplant cancers may be improving in recent years, these results suggest there are deficiencies in monitoring and reporting by transplant centers. Research use of SRTR cancer data may lead to inaccurate results and, in particular, an underestimation of cancer risk.
To cite this abstract in AMA style:
Engels E, Segev D, Snyder J, Kasiske B, Yanik E. Discrepancies Between the U.S. Transplant Registry and Cancer Registries Regarding Cancer Diagnoses in Transplant Recipients [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/discrepancies-between-the-u-s-transplant-registry-and-cancer-registries-regarding-cancer-diagnoses-in-transplant-recipients/. Accessed June 2, 2026.« Back to 2015 American Transplant Congress
