Development of Optimal Simplified Equation for Estimation of Mycophenolic Acid Exposure Following Enteric-Coated Mycophenolate Sodium Administration in Chinese Renal Transplant Recipients
MyERA Study Group, Hangzhou, China.
Meeting: 2018 American Transplant Congress
Abstract number: C98
Keywords: Area-under-curve (AUC), Immunosuppression, Kidney transplantation, Pharmacokinetics
Session Information
Session Name: Poster Session C: Kidney Immunosuppression: Novel Regimens and Drug Minimization
Session Type: Poster Session
Date: Monday, June 4, 2018
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall 4EF
Background: Limited sampling strategies (LSS) aids in overcoming the complexity of extensive PK sampling and supporting the calculation of mycophenolic acid (MPA) exposure. This study aims to select sparse and appropriate time points for PK sampling, and develop optimal simplified equation (OSE) by using LSS for estimation of accurate MPA exposure in Chinese renal transplant recipients (RTR) receiving organs from cardiac death donors.
Methods: This open-label, multicenter, prospective Phase IV observational study enrolled 250 RTR, receiving standard (s; 1440 mg/d) or high (h; >1440 mg/d) dose EC-MPS with calcineurin inhibitors (CNI: cyclosporine, CsA or tacrolimus, TAC) across 19 centers. The PK sampling time points with evaluable concentrations in >80% RTR (N=116; at 0, 0.5, 1, 1.5, 2, 3, 4, 6, 8, 10, and 12h) were selected for evaluation. An OSE of MPA AUC0-12h was developed using multiple linear regression model by stepwise screening, with full AUC0-12h as dependent variable and serum MPA AUC0-12h at each time point as independent variable.
Results: The time points selected in the OSE were 0.5, 2, 4, and 8h for sEC-MPS+CsA and sEC-MPS+TAC, and 1.5, 3 and 6h for hEC-MPS+TAC. The corresponding coefficients of determination (r2) were ≥0.9 (P<0.05), and mean absolute prediction errors (APE) <15% (Tables 1a&1b). In addition, an exploratory analysis based on the PK profile, and feasibility of clinical practice, revealed that time points of 0.5, 2, and 8h were also suitable for hEC-MPS+TAC arm (20.92+0.47C0.5+0.99C2+11.45C8;r2=0.858; P<0.001; APE%=14.4). Results from exploratory and statistical analysis were consistent for sEC-MPS+CsA and sEC-MPS+TAC.
Conclusions: OSE were recommended using both statistical and exploratory analysis. The OSE based on exploratory analysis is more convenient for clinical application as it is regardless of initial EC-MPS dose and CNI used in Chinese population. However, unbalanced sample size across arms necessitates further validation. 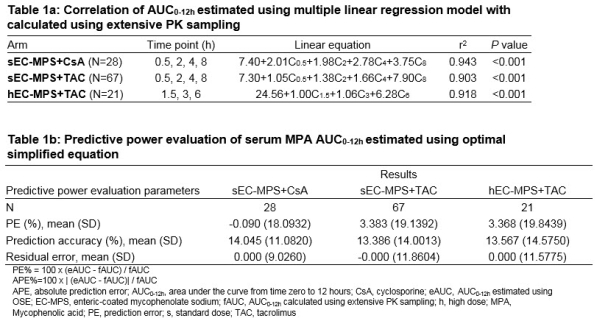
CITATION INFORMATION: Peng W., Tian C., Chen Z., Xue W., Wang G., Ye Q., Zhang X., Gu M., Zhao M., Wang C., Huang C., Yan J., Zhang W., Ding C., Fu Y., Ye S., Hu X., Tan R., Li M., Deng R., Ke Y., Wu W., Fan M., Chen J. Development of Optimal Simplified Equation for Estimation of Mycophenolic Acid Exposure Following Enteric-Coated Mycophenolate Sodium Administration in Chinese Renal Transplant Recipients Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Peng W, Tian C, Chen Z, Xue W, Wang G, Ye Q, Zhang X, Gu M, Zhao M, Wang C, Huang C, Yan J, Zhang W, Ding C, Fu Y, Ye S, Hu X, Tan R, Li M, Deng R, Ke Y, Wu W, Fan M, Chen J. Development of Optimal Simplified Equation for Estimation of Mycophenolic Acid Exposure Following Enteric-Coated Mycophenolate Sodium Administration in Chinese Renal Transplant Recipients [abstract]. https://atcmeetingabstracts.com/abstract/development-of-optimal-simplified-equation-for-estimation-of-mycophenolic-acid-exposure-following-enteric-coated-mycophenolate-sodium-administration-in-chinese-renal-transplant-recipients/. Accessed May 30, 2026.« Back to 2018 American Transplant Congress
