Delayed Graft Function in Pediatric Living Donor Kidney Transplantation
Surgery, UT Southwestern Medical Center, Dallas, TX
Meeting: 2021 American Transplant Congress
Abstract number: 82
Keywords: Graft survival, Kidney, Outcome, Pediatric
Topic: Clinical Science » Kidney » Kidney: Pediatrics
Session Information
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:35pm-6:40pm
Presentation Time: 6:35pm-6:40pm
Location: Virtual
*Purpose: Pediatric recipients of living donor kidneys have a low rate of delayed graft function (DGF). The aim of this study was to examine the incidence, risk factors and outcomes of DGF in pediatric patients who received a living donor allograft.
*Methods: The UNOS database was queried to examine all pediatric patients transplanted with a living donor kidney between 2000—2020. A pediatric recipient was defined as one who was transplanted prior to the age of 18 years. Donor and recipient demographic data were examined, as were survival and outcomes. DGF was defined as the need for dialysis within the first week after transplant. A p-value of <0.05 was considered to be significant.
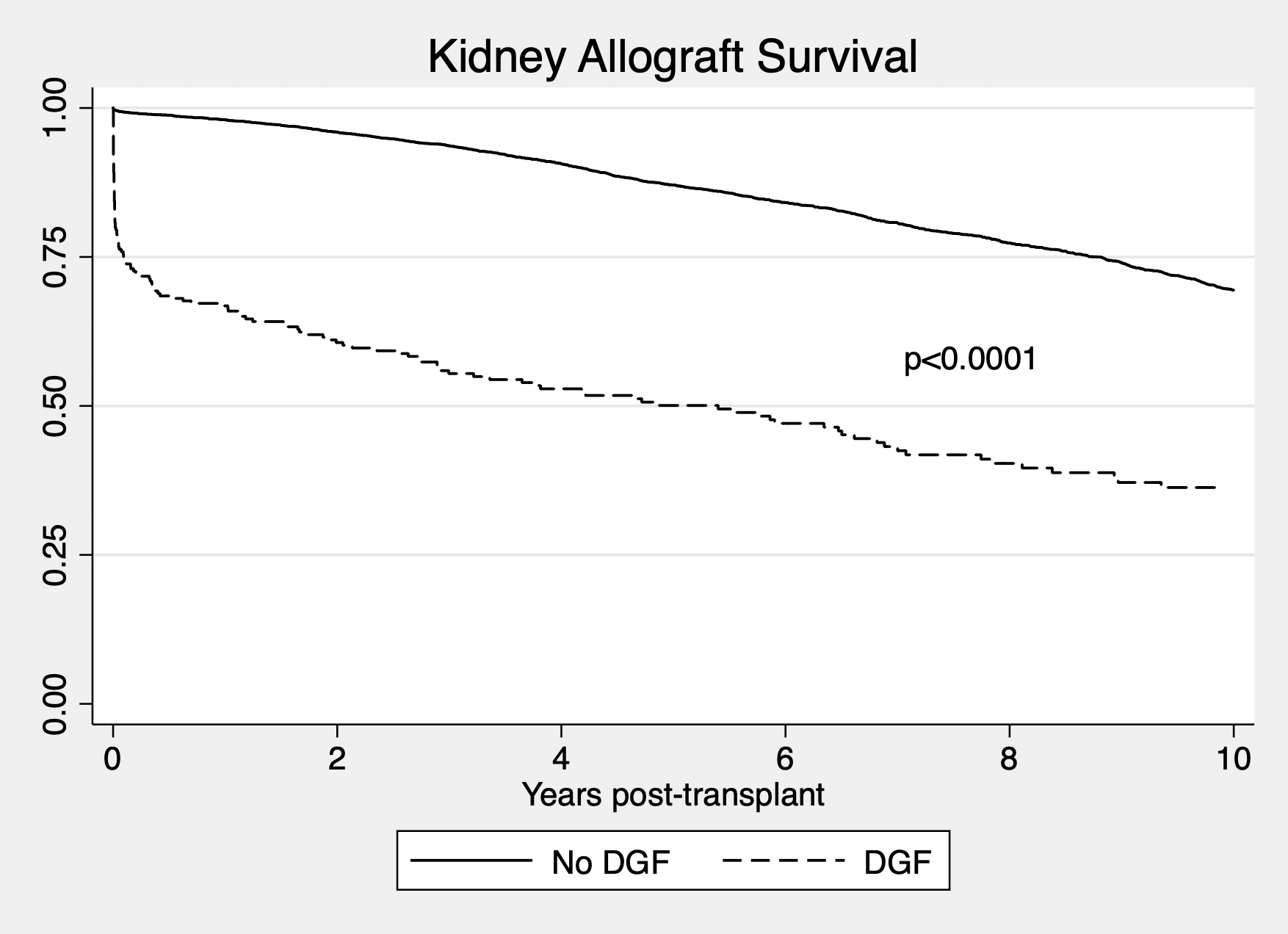
*Results: 6,480 pediatric patients received a living donor (LD) kidney transplant during the study period. 269 (4.2%) developed DGF post-transplant. Donors were similar in age DGF and control groups (37.2 vs. 37.0 yrs), had similar preoperative creatinine (0.86 mg/dL in both), ethnicity, and gender, but donor BMI was higher in the DGF group (27.6 vs. 26.8 kg/m2, p=0.004). Cold ischemia time (2.4 vs. 2.3 h) was also similar. Recipients of kidneys with DGF were similar in age (9.8 vs. 10.1 y), and had higher BMI (20.3 vs. 19.4 kg/m2, p=0.002). Initial and final cPRAs were similar (2.2 vs. 2.4% initial; 10.5 vs. 8.9% final). HLA mismatch was similar in both groups (3.0 vs. 2.8). Focal segmental glomerulosclerosis (FSGS) was the most common diagnosis in recipients with DGF (23%) and was significantly more frequent in the DGF group compared to control (23% vs. 10%, p=0.001). Small recipients (weight < 15 kg) were found to have a significantly higher rate of DGF (24.9 vs. 19.8%, p=0.04). Length of stay for recipients with DGF was twice that of the control group (23.4 vs. 10.1 d, p<0.0001). Patients with DGF had higher rates of rejection at 6 and 12 months post transplant (24.8 vs. 8.0% at 6 months, p<0.0001; 26.4 vs. 11.6% at 12 months, p<0.0001). Recipients of LD kidneys who developed DGF had significantly worse allograft survival when compared to control (Figure 1, p<0.0001) with 1 year graft survival over 30% lower than control (67% vs. 98%). The most common causes of allograft loss were graft thrombosis (25.3%), rejection (17.5%), and recurrent disease (15.6%).
*Conclusions: Pediatric living donor kidney transplant recipients who experience DGF have significantly poorer allograft survival. Immunologic events, recurrent disease and technical complications appear to underlie these poor outcomes and should be considered especially in younger recipients with FSGS. Optimizing the donor recipient combination to avoid compounding risks should allow for better outcomes.
To cite this abstract in AMA style:
MacConmara M, Shah J, Gregorio Lde, Desai D, Vagefi P, Hwang CS. Delayed Graft Function in Pediatric Living Donor Kidney Transplantation [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/delayed-graft-function-in-pediatric-living-donor-kidney-transplantation/. Accessed May 21, 2026.« Back to 2021 American Transplant Congress