A Specific Inhibitor of C1s, TNT003, Prevents Complement-Mediated Enhancement of Monocyte Adhesion to Endothelium in Response to HLA Antibodies
1Immunogenetics Center, UCLA, Los Angeles, CA
2Leiden University, Leiden, Netherlands
3True North Therapeutics, San Francisco, CA.
Meeting: 2015 American Transplant Congress
Abstract number: D78
Keywords: Alloantibodies, Endothelial activation, HLA antibodies, Mononuclear leukocytes
Session Information
Session Name: Poster Session D: Innate Immunity in Transplantation
Session Type: Poster Session
Date: Tuesday, May 5, 2015
Session Time: 5:30pm-6:30pm
 Presentation Time: 5:30pm-6:30pm
Presentation Time: 5:30pm-6:30pm
Location: Exhibit Hall E
HLA antibody (Ab) mediated complement activation on endothelial cells (EC) is likely a critical driver of antibody-mediated inflammation. Production of complement anaphylatoxins by HLA antibodies (Ab) can induce EC activation and recruit leukocytes, leading to graft injury during AMR. We hypothesized that when C3a and/or C5a are generated by HLA Ab, EC activation and resultant monocyte recruitment would be enhanced compared with HLA Ab alone.
Primary human aortic EC were stimulated with chimeric pan-HLA I IgG1, human allele-specific monoclonal Ab, or allosera, in the presence of 25% normal human serum complement (C') or heat-inactivated C' (HI-C'), and/or purified C3a/C5a. EC activation was measured by detection of cell surface P-selectin, and binding of a monocytic cell line Mono Mac 6 (MM6). C' was preincubated with 1-25μg/mL of TNT003, an anti-C1s Ab, to inhibit the classical complement pathway.
In the presence of C' (but not HI-C'), C3a and C5a were increased by 2- and 3-fold in the supernatants of EC treated with HLA Ab, but not negative serum without HLA Ab (NS). Stimulation of EC with purified C5a but not C3a rapidly increased cell surface P-selectin and adhesion of MM6. Direct activation of MM6 with C3a or C5a increased adhesion to EC in a concentration dependent manner. Stimulation of EC with both HLA Ab and C3a or C5a additively increased P-selectin expression and adherent MM6. P-selectin and MM6 binding were significantly enhanced when cells were stimulated with HLA Ab in C' compared with HI-C' and C1q-depleted C', confirming the role of the classical pathway.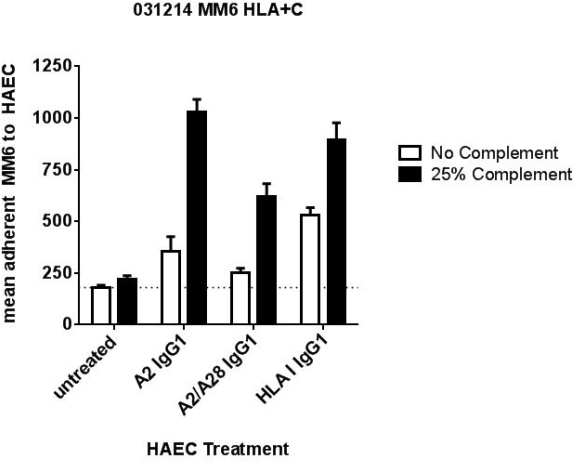 Finally, pretreatment of C' with TNT003 inhibited enhancement of MM6 adhesion in a dose-dependent manner.
Finally, pretreatment of C' with TNT003 inhibited enhancement of MM6 adhesion in a dose-dependent manner.
These findings demonstrate a mechanism by which complement and HLA Ab synergize to promote recruitment of recipient immune cells during AMR. Proximal antagonism of the classical complement cascade may prove superior to terminal complement inhibition by preventing production of anaphylatoxins C3a as well as C5a.
To cite this abstract in AMA style:
Valenzuela N, Thomas K, Mulder A, Parry G, Parker S, Reed E. A Specific Inhibitor of C1s, TNT003, Prevents Complement-Mediated Enhancement of Monocyte Adhesion to Endothelium in Response to HLA Antibodies [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/a-specific-inhibitor-of-c1s-tnt003-prevents-complement-mediated-enhancement-of-monocyte-adhesion-to-endothelium-in-response-to-hla-antibodies/. Accessed July 12, 2025.« Back to 2015 American Transplant Congress
