A Comparison of Plasmapheresis Methods in the Treatment of Late Antibody Mediated Rejection
1Saint Louis University, Saint Louis, MO, 2Division of Nephrology, Istanbul University, Istanbul, Turkey, 3Department of Pathology, Istanbul University, Istanbul, Turkey, 4Division of Hematology, Istanbul University, Istanbul, Turkey
Meeting: 2021 American Transplant Congress
Abstract number: 453
Keywords: Antibodies, Graft survival, Plasmapheresis, Rejection
Topic: Clinical Science » Kidney » Kidney Chronic Antibody Mediated Rejection
Session Information
Session Name: Kidney Alloimmune Responses
Session Type: Poster Video Chat
Date: Monday, June 7, 2021
Session Time: 7:30pm-8:30pm
 Presentation Time: 8:00pm-8:10pm
Presentation Time: 8:00pm-8:10pm
Location: Virtual
*Purpose: Effective management to prevent allograft loss after late antibody mediated rejection (ABMR) in kidney transplant (KTx) recipients remains a vexing clinical challenge. While plasma exchange (PE) with IVIG has been proposed as a useful treatment, alternatives such as double filtration plasmapheresis (DFPP) and immunoadsorption (IA) represent promising strategies for antibody depletion. We compared graft outcomes associated with use of PE, DFPP and IA in the treatment of late ABMR.
*Methods: We performed a retrospective cohort study of 69 KTx recipients [living donor (n=54, 79%), deceased donor (n=14, 21%)] who were diagnosed with biopsy-confirmed “late” (>1 year post-KTx) ABMR at one center and underwent therapeutic plasmapheresis. ABMR was identified at a median of 7.9 (IQR, 3-13) years after KTx. Therapeutic plasmapheresis modalities included: PE (n=31), DFPP (n=22) or IA with protein A (n=16). The primary outcome was allograft loss, defined as return to dialysis or re-transplantation. Multivariate Cox proportional survival analysis was conducted with covariate adjustments to identify predictors of allograft loss.
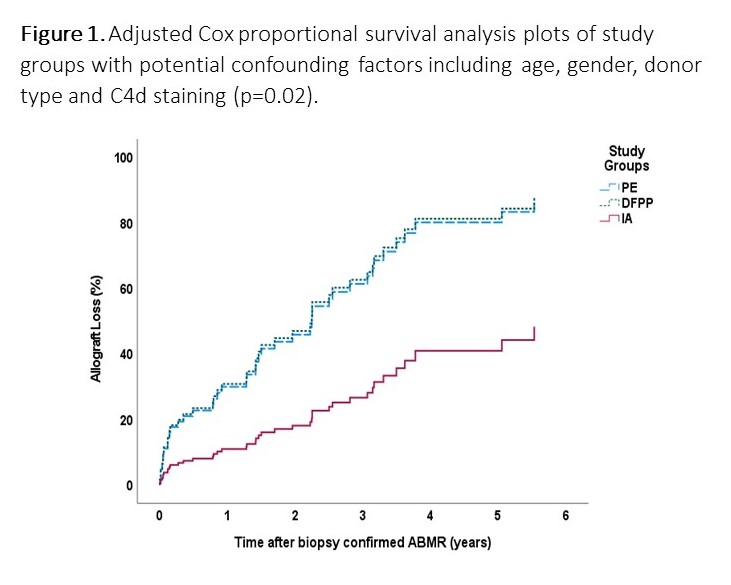
*Results: The study groups were similar regarding age, gender, donor type, donor age and gender, eGFR at ABMR diagnosis and time from KTx to ABMR. Donor specific antibody (DSA) was positive in 84% of PE, 91% of DPFF and 94% of IA groups (p=0.55), and C4d staining in peritubular capillaries was negative in 36%, 23% and 56% (p=0.001) of study groups, respectively. Patients received a total dose of 2 g/kg IVIG with (75%) or without (25%) rituximab (375 mg/m2). There were no differences regarding IVIG alone or IVIG with rituximab treatment rates in all groups (p=0.16). Adjusted Cox proportional survival analysis with potential confounding factors including age, gender, donor type and C4d staining showed that IA was significantly associated with 67% reduced risk of allograft loss (adjusted hazard ratio 0.120.330.87)(p=0.02). (Fig 1)
*Conclusions: While limited by small sample size, this pilot study suggests the efficacy of IA in the treatment of late ABMR. Future prospective and controlled studies should examine the robustness of this observation.
To cite this abstract in AMA style:
Caliskan Y, Yazici H, Dirim AB, Aksoy E, Safak S, Garayeva N, Mirioglu S, Yegit O, Oto OA, Ozluk Y, Besisik S, Turkmen A, Lentine K. A Comparison of Plasmapheresis Methods in the Treatment of Late Antibody Mediated Rejection [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/a-comparison-of-plasmapheresis-methods-in-the-treatment-of-late-antibody-mediated-rejection/. Accessed May 18, 2026.« Back to 2021 American Transplant Congress