Variation in Co-Medication Use According to Kidney Transplant Immunosuppressive Regimen: Application of Integrated Registry and Pharmacy Claims Data.
K. Lentine,1 A. Naik,2 M. Schnitzler,1 D. Axelrod,3 J. Chen,1 D. Brennan,4 D. Segev,5 B. Kasiske,6 H. Randall,1 V. Dharnidharka.4
1Saint Louis Univ, St. Louis
2Univ Michigan, Detroit
3Dartmouth Univ, Hanover
4Washington Univ, St. Louis
5Johns Hopkins Univ, Baltimore
6Univ Minnesota, Minneapolis.
Meeting: 2016 American Transplant Congress
Abstract number: 336
Keywords: Immunosuppression, Infection, Kidney transplantation, Metabolic complications
Session Information
Session Name: Concurrent Session: Medication Errors, Variability and Adherence
Session Type: Concurrent Session
Date: Monday, June 13, 2016
Session Time: 4:30pm-6:00pm
 Presentation Time: 5:18pm-5:30pm
Presentation Time: 5:18pm-5:30pm
Location: Room 302
Background: While modern immunosuppressive therapies (ISx) have substantially reduced acute rejection, ISx medications have many side effects, and transplant recipients must take an array of “co-medications” to help mitigate complications. Co-medication utilization patterns are not well described in large, representative samples due to lack of available data.
Methods: We integrated national U.S. transplant registry data with pharmacy records (2005-2010) from a large pharmaceutical claims clearinghouse to examine treatments for anemia, metabolic disorders and infections according to ISx regimen in mo 6-12 post-transplant (N=22,453). Associations of ISx with co-medication use (adjusted odds ratio, aOR) were examined with multivariate logistic regression including adjustment for recipient, donor and transplant factors.
Results: Compared to a reference regimen of tacrolimus (Tac), mycophenolate (MPA) and prednisone, sirolimus-based ISx was associated with significantly (P<0.05) higher use of ESAs (aOR 2.52), iron (aOR 2.26), statins (aOR 1.47), fibrates (aOR 2.35), and phosphorous binders (aOR 2.85) (Figure). Patterns were similar after adjustment for first-year estimated glomerular filtration rate. Cyclosporine-based ISx was associated with more common use of anemia treatments. Compared to those taking triple ISx, recipients of tacrolimus-based dual and mono-therapies had lower use of statins, ACEi/ARBs, and anti-bacterial agents. Recipients of steroid-free ISx were less commonly treated for new onset diabetes.
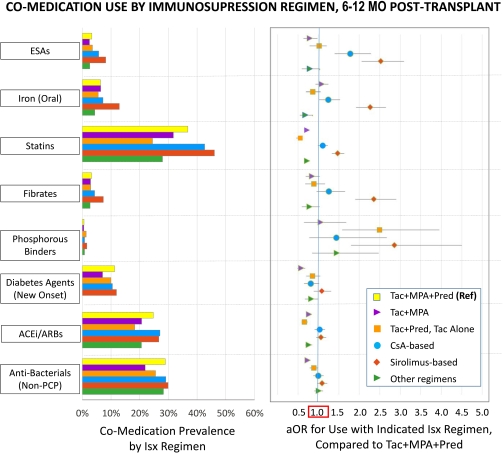
Conclusions: Alternate ISx regimens are associated with varying treatment requirements for hematologic, metabolic and infectious complications. Co-medication use should be considered in the cost-effectiveness and individualization of ISx regimens.
CITATION INFORMATION: Lentine K, Naik A, Schnitzler M, Axelrod D, Chen J, Brennan D, Segev D, Kasiske B, Randall H, Dharnidharka V. Variation in Co-Medication Use According to Kidney Transplant Immunosuppressive Regimen: Application of Integrated Registry and Pharmacy Claims Data. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Lentine K, Naik A, Schnitzler M, Axelrod D, Chen J, Brennan D, Segev D, Kasiske B, Randall H, Dharnidharka V. Variation in Co-Medication Use According to Kidney Transplant Immunosuppressive Regimen: Application of Integrated Registry and Pharmacy Claims Data. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/variation-in-co-medication-use-according-to-kidney-transplant-immunosuppressive-regimen-application-of-integrated-registry-and-pharmacy-claims-data/. Accessed June 1, 2026.« Back to 2016 American Transplant Congress
