Using Nudges for Liver Transplantation Prehabilitation: Results from PRIMER (Prehabilitation Intervention to Maximize Early Recovery), a Home-Based Behavioral Pilot and Feasibility Trial
University of Pennsylvania, Philadelphia, PA
Meeting: 2020 American Transplant Congress
Abstract number: A-114
Keywords: Kidney/liver transplantation, Liver cirrhosis
Session Information
Session Name: Poster Session A: Liver: Portal Hypertension and Other Complications of Cirrhosis
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
*Purpose: Data on effective prerehabilitation in liver transplant (LT) is limited. We studied the feasibility and acceptability of a pilot randomized controlled trial of a 12-week home-based prehabilitation exercise intervention for LTCs.
*Methods: We randomized 30 LTCs (n=20 intervention, n=10 control) on the LT waitlist or in active LT evaluation at a large transplant center. Inclusion criteria were at least one the following: 1) impaired physical function measured by Short Physical Performance Battery (SPPB), 2) weakness in handgrip testing, or 3) moderate or severe malnutrition. Patients with MELD 25 or greater, no smartphones, a hospital admission in the last 30 days, or those already receiving physical therapy were excluded. All participants received personal fitness trackers and received general dietary and exercise recommendations. The intervention arm additionally received text messages with 1) daily step targets increasing by 15% every 2 weeks, 2) tailored exercise videos, and 3) twice daily medication adherence reminders. The study coordinator conducted weekly telephone check-ins with patients to assess intervention fidelity. Daily financial incentives were provided for adherence to daily step goals and weekly check-ins (2 USD per day for meeting step targets, 9 USD per weekly check-in). Primary study outcomes were feasibility and acceptability; mean daily steps the last 2 weeks of the study was an exploratory outcome.
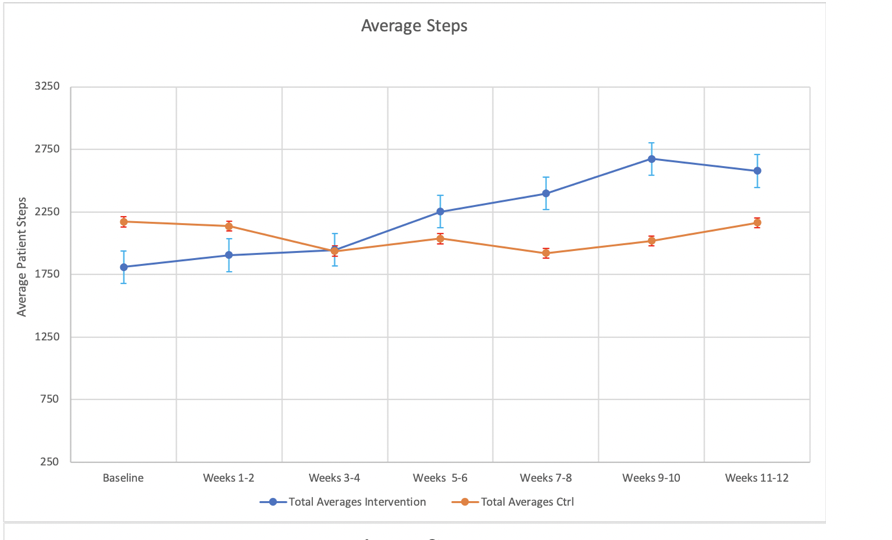
*Results: The median MELD was 13 [IQR 11-16]. A total of 9 (30%) were frail by grip strength; 20 (67%) had history of hepatic encephalopathy, 6 (20%) had previous spontaneous bacterial peritonitis, 9 (30%) had diabetes. The mean baseline BMI was 28 kg/m2 (SD 5.7). There were no significant differences in baseline characteristics by study arm. A total of 28/30 patients completed the study and adherence to weekly check-ins was 86%.The mean step count at the last 2 weeks of the study was 656 steps higher in the intervention arm than in the control arm (intervention: 2674 (SD 1498), control: 2018 (SD=1517), p=0003).
*Conclusions: A home-based, low-cost physical activity and prehabilitation intervention for LTCs not meeting standard physical therapy criteria was feasible with high retention and resulted in increased daily walking warranting larger, prospective trials.
To cite this abstract in AMA style:
Serper M, Jones LS, Clement T, Reddy K, Duarte-Rojo A, Reese PP. Using Nudges for Liver Transplantation Prehabilitation: Results from PRIMER (Prehabilitation Intervention to Maximize Early Recovery), a Home-Based Behavioral Pilot and Feasibility Trial [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/using-nudges-for-liver-transplantation-prehabilitation-results-from-primer-prehabilitation-intervention-to-maximize-early-recovery-a-home-based-behavioral-pilot-and-feasibility-trial/. Accessed May 17, 2026.« Back to 2020 American Transplant Congress