Tolerance Induction by αLFA1 Monotherapy Reveals Organ Specific CD8-Dependent Immune Mechanisms
CCTCARE, Dept. of Surgery, University of Colorado Denver, Aurora, CO
Pulmonary Sciences and Critical Care Medicine, University of Colorado Denver, Aurora, CO
Cardiology, Dept. of Pediatrics, The Colorado Children's Hospital, Aurora, CO
Meeting: 2013 American Transplant Congress
Abstract number: 79
Purpose: Innate and adaptive immunity requirements for tolerance induction appear to be agent and organ specific. Based on our previously published data for tolerance requirements in islet allografts, we sought to investigate whether tolerance induction to cardiac allografts utilizing ΑLFA-1 mAb monotherapy had similar requirements.
Methods: BALB/c or C3H hearts were transplanted into B6, B6rag-/-, Β2M or CD1-/- mice and were untreated or treated with ΑLFA-1 or control rat IgG or ΑNK1.1 or ΑCD8 or adoptively transferred with tolerized or naive splenocytes.
Results: Untreated or IgG treated B6 mice reject their BALB/c cardiac grafts acutely (8.8 and 10.7 days) while ΑLFA-1 treated B6 recipients do not (95.6±18.6 days). Mice with allograft survival >100 days were shown to have transferable donor specific tolerance with grafts exposed to tolerized splenocytes surviving >100 days in B6rag-/- recipients of BALB/c hearts, whereas C3H hearts rejected acutely in 6.5 days. Transferred naive cells caused acute rejection. To determine if NK cells were required for tolerance B6 recipients were treated with both ΑLFA-1 and ΑNK1.1 and survived >100 days. Untreated CD1-/- recipients (devoid of non-classical MHC class I expression restricted NKT cells) acutely rejected allografts while ΑLFA-1 treated grafts survived >100 days. This survival persisted when ΑNK1.1 was used to deplete NK cells. On the contrary, host MHC class I and CD8 T cells do contribute to tolerance induction.
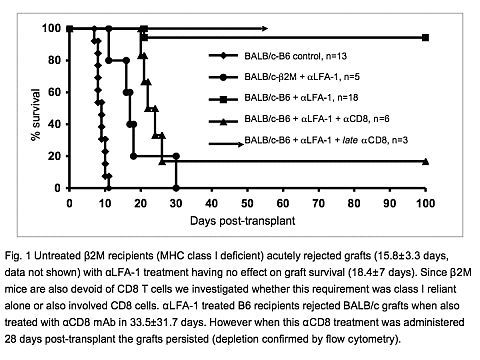
Conclusion: We find that ΑLFA-1 mAb constitutes a potent tolerance-inducing monotherapy in fully vascularized cardiac allografts. Unlike islet allografts, NK cells and NKT cells are not required for induction of ΑLFA-1 tolerance to cardiac allografts. Further, host MHC class I expression is necessary, and more importantly, CD8 T cells are critical for the induction of tolerance, but appear not to be required for the maintenance of that tolerance.
To cite this abstract in AMA style:
Plenter R, Grazia T, Zamora M, Lim C, Gill R, Pietra B. Tolerance Induction by αLFA1 Monotherapy Reveals Organ Specific CD8-Dependent Immune Mechanisms [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/tolerance-induction-by-lfa1-monotherapy-reveals-organ-specific-cd8-dependent-immune-mechanisms/. Accessed May 24, 2026.« Back to 2013 American Transplant Congress
