The THINKER Trial: Quality of Life Results at 6- and 12-Month Follow-Up among Hepatitis C-Negative Patients Who Received Kidney Transplants from Deceased Donors with Hepatitis C Infection
V. Potluri, D. Goldberg, A. Peter, B. Emily, B. Roy, S. Deirdre, M. Levine, J. Trofe-Clark, M. Bleicher, S. Goral, A. Sicilia, S. Nazarian, R. Reddy, P. Porrett, P. Reese.
Univ of Pennsylvania, Philadelphia, PA.
Meeting: 2018 American Transplant Congress
Abstract number: 44
Keywords: Donors, Kidney transplantation, marginal, Outcome
Session Information
Session Name: Concurrent Session: Kidney Infectious - Viral Hepatitis
Session Type: Concurrent Session
Date: Sunday, June 3, 2018
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:18pm-3:30pm
Presentation Time: 3:18pm-3:30pm
Location: Room 4C-4
Background: Short-term (6-month) data from the first 10 subjects in the pilot THINKER-1 trial (NCT02743897; funder: Merck) suggested that kidneys from HCV-infected donors could be safely transplanted into HCV-negative patients. However, nothing is known about quality of life outcomes for these patients, who may have a different transplant experience due to concerns about stigma or the risks of infection or treatment. We now report 12-month quality of life data on the 10 THINKER-1 recipients as well as 6-month data on 10 recipients from an expanded cohort (THINKER-2).
Methods: Participants completed the RAND-36 questionnaire – a widely validated quality of life instrument – at enrollment and at regular intervals post-transplant. The Physical Component Summary (PCS) score and Mental Component Summary (MCS) score are normalized to population standards, with higher scores indicating better quality of life.
Results: Median age was 58 years (IQR 50 – 62), 70% were male and 35% were self-reported black race. Normalized mean PCS scores declined at 4 weeks and then increased steadily; the mean PCS improvement from pre-transplant to 12-months was 8.6. Normalized mean MCS scores also declined at 4 weeks, however, the MCS scores subsequently returned to baseline and remained stable at the end of 12 months. The Figure shows trajectories over time in individual quality of life domains as well as the PCS and MCS.
In conclusion, quality of life remains generally stable or improves after transplantation using HCV-infected kidneys for uninfected recipients. Research is ongoing to determine specific reasons for these trends in quality of life. These findings provide additional evidence that transplantation with HCV-infected kidneys, followed by treatment, provides benefit to patients with end-stage renal disease. 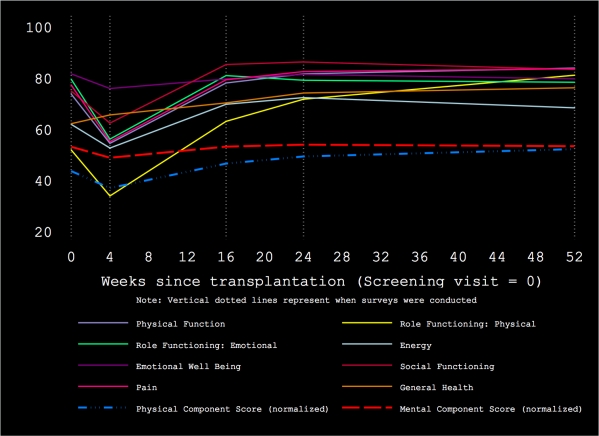
CITATION INFORMATION: Potluri V., Goldberg D., Peter A., Emily B., Roy B., Deirdre S., Levine M., Trofe-Clark J., Bleicher M., Goral S., Sicilia A., Nazarian S., Reddy R., Porrett P., Reese P. The THINKER Trial: Quality of Life Results at 6- and 12-Month Follow-Up among Hepatitis C-Negative Patients Who Received Kidney Transplants from Deceased Donors with Hepatitis C Infection Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Potluri V, Goldberg D, Peter A, Emily B, Roy B, Deirdre S, Levine M, Trofe-Clark J, Bleicher M, Goral S, Sicilia A, Nazarian S, Reddy R, Porrett P, Reese P. The THINKER Trial: Quality of Life Results at 6- and 12-Month Follow-Up among Hepatitis C-Negative Patients Who Received Kidney Transplants from Deceased Donors with Hepatitis C Infection [abstract]. https://atcmeetingabstracts.com/abstract/the-thinker-trial-quality-of-life-results-at-6-and-12-month-follow-up-among-hepatitis-c-negative-patients-who-received-kidney-transplants-from-deceased-donors-with-hepatitis-c-infection/. Accessed May 16, 2026.« Back to 2018 American Transplant Congress
