Safer and More Effective Treatment of Acute Postoperative Pain in Living Liver Donors
D. Ladner,1 R. Fisher,2 J. Guarrera,3 E. Pomfret,4 M. Simpson,4 A. Daud,5 T. Curtis,5 E. Reyes,5 A. Skaro,1 D. Woods.5
1Northwestern University, Chicago, IL
2Virginia Commonwealth University, Richmond, VA
3Columbia University, New York, NY
4Lahey Clinic, Boston, MA
5CHS, Northwestern University, Chicago, IL.
Meeting: 2015 American Transplant Congress
Abstract number: B154
Keywords: Living-related liver donors, Morbidity, Outcome, Pain
Session Information
Session Name: Poster Session B: Liver: Living Donors
Session Type: Poster Session
Date: Sunday, May 3, 2015
Session Time: 5:30pm-6:30pm
 Presentation Time: 5:30pm-6:30pm
Presentation Time: 5:30pm-6:30pm
Location: Exhibit Hall E
BACKGROUND: As part of Adult-to-Adult Living Donor Liver Transplant Cohort Study (A2ALL), donors were found to experience significant pain post donation, and more than half of the donors experienced opioid related complications associated with standard of care (SOC). A multimodal opioid-sparing postoperative donor pain protocol was developed. Preliminary data are reported (enrollment concludes 5/15)
METHOD: The pain protocol was instituted 2/2013 at 4 A2ALL centers. A trained RN reviewed the medical records of every complication. Baseline data (PRE) were compared to patients who received the pain protocol (POST) and verified by an independent monitor.
RESULTS: 90 donors received SOC, and 23 the pain protocol. The most frequent complications associated with opioid usage were nausea (61%), hypoxia (56%), constipation (48%) and hypotension (46%). All opioid related complications diminished POST. Complications associated with inadequate pain control were reduced or unchanged (tachycardia, hypertension, tachypnea). Pain scores were similar PRE and POST.
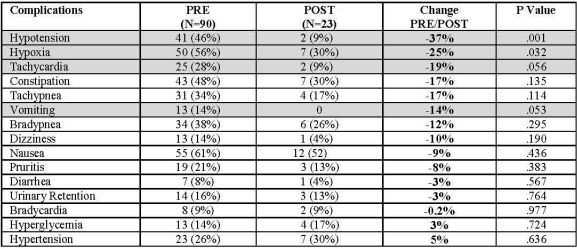
CONCLUSIONS: Opioid sparing pain management can effectively reduce the high rate of complications related to opioid use without worsening pain perception in living liver donors. The preliminary data suggest that the opioid sparing pain protocol is significantly safer than the present SOC.
To cite this abstract in AMA style:
Ladner D, Fisher R, Guarrera J, Pomfret E, Simpson M, Daud A, Curtis T, Reyes E, Skaro A, Woods D. Safer and More Effective Treatment of Acute Postoperative Pain in Living Liver Donors [abstract]. Am J Transplant. 2015; 15 (suppl 3). https://atcmeetingabstracts.com/abstract/safer-and-more-effective-treatment-of-acute-postoperative-pain-in-living-liver-donors/. Accessed May 25, 2026.« Back to 2015 American Transplant Congress
