Recovery of eGFR in Both Donors and Recipients After Living Donor Kidney Transplantation.
MedStar Georgetown Transplant Institute, Washington, DC.
Meeting: 2016 American Transplant Congress
Abstract number: A139
Keywords: Donation, Glomerular filtration rate (GFR), Graft function, Kidney transplantation
Session Information
Session Name: Poster Session A: Kidney Donor Outcomes
Session Type: Poster Session
Date: Saturday, June 11, 2016
Session Time: 5:30pm-7:30pm
 Presentation Time: 5:30pm-7:30pm
Presentation Time: 5:30pm-7:30pm
Location: Halls C&D
Purpose: LD kidney donation provides a much needed resource for patients with end stage renal disease. Providing adequate renal mass to the recipient (R) is needed, but equally as important is leaving sufficient long-term renal function for donor (D). Based on experience, but without much hard data, most centers use a cutoff for donor eGFR of 80 mL/min. With compensatory hypertrophy, this is estimated to leave D with roughly 70% of pre-donation function. Hypertrophy in Rs is less clear, in the setting of vasoconstrictors and nephrotoxic immunosuppression. We examined the degree of function/compensatory hypertrophy in both D and R post-transplant.
Methods: Demographics, preop eGFR, and eGFR 6 months postop (6M) were reviewed. eGFR was calculated using MDRD-4 equation. Statistics performed using SPSS.
Results:64 sequential pairs (n=128 patients) were studied. There were more male Rs than Ds (56 vs 34%), and Rs were slightly older than Ds (50.8 vs 46.8 years). There was no difference between R and D BMI (27.2 vs 26.4 kg/m2). Theoretically, both D and R should start with 50% of D's preop eGFR; this value was used as baseline for both pts. Average D preop eGFR was 100.4+/-23 mL/min. Mean D eGFR at 6M was 58.31 mL/min, and mean R eGFR was 59.45 mL/min (p=0.634). There was an average of 15.7% increase in D eGFR and 17.9% increase in R eGFR (p=NS). There were no graft losses in the cohort, and separate analysis showed outliers had no significant effect on results. When donors were divided into 4 quartiles based on preop eGFR, all showed similar gains in eGFR except at the highest level where some patients actually regressed slightly. 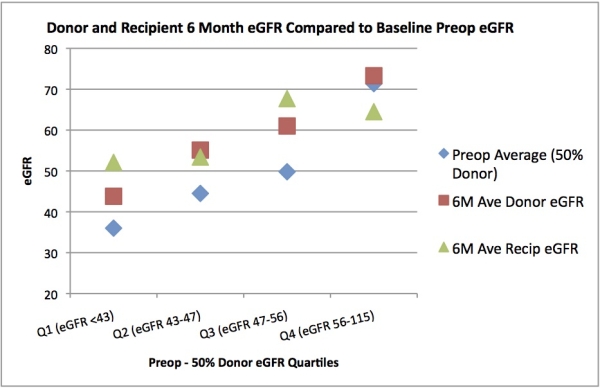
Conclusions: Our results suggest that both D and R kidneys undergo compensatory hypertrophy at roughly similar rates, approximately 17% over estimated baseline. Of note, Rs have significant hypertrophy despite use of nephrotoxic and vasoconstrictive agents post-transplant. Understanding the behavior of eGFR in both Ds and Rs allows us to more accurately select donors who may have otherwise been excluded, while maintaining their long-term safety.
CITATION INFORMATION: Verbesey J, Gilbert A, Li A, Reissmann M, Foreman N, Grafals M, Javaid B, Abrams P, Ghasemian S, Cooper M. Recovery of eGFR in Both Donors and Recipients After Living Donor Kidney Transplantation. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Verbesey J, Gilbert A, Li A, Reissmann M, Foreman N, Grafals M, Javaid B, Abrams P, Ghasemian S, Cooper M. Recovery of eGFR in Both Donors and Recipients After Living Donor Kidney Transplantation. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/recovery-of-egfr-in-both-donors-and-recipients-after-living-donor-kidney-transplantation/. Accessed July 12, 2025.« Back to 2016 American Transplant Congress
