Probability of a Positive AHG-CDC Crossmatch Is Determined by the Number, Strength, and Specificity of Donor-Specific Antibodies
Division of Nephrology, Department of Medicine, University of British Columbia, Vancouver, BC, Canada
Meeting: 2013 American Transplant Congress
Abstract number: 411
Background: The factors determining the relationship between donor-specific antibodies (DSA) and the anti-human globulin complement-dependent cytotoxicity crossmatch (AHG-CDC XM) are not clearly defined. Canadian consensus guidelines propose an MFI of 1000 as the threshold for identification of anti-HLA antibodies via Luminex technology, but we hypothesize that its relationship to AHG-CDC may depend on the number, strength, and specificity of DSA present in recipient serum.
Methods: Class I and II anti-HLA antibody specificities in 220 serum samples from 120 PRA-positive patients wait-listed for kidney transplantation were determined by single-antigen testing via Luminex, and expressed as the mean florescent intensity (MFI). AHG-CDC T-cell XM was performed on all samples and the results correlated by logistic regression modeling and receiver operator curve (ROC) analysis.
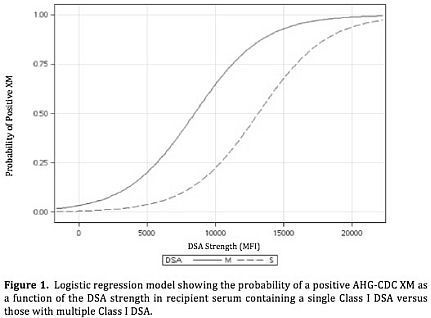
Results: 38/220 samples (17%) contained only Class II DSA: 0% (0/38) were associated with a positive AHG-CDC, independent of the MFI value or the number of DSA. 73/220 samples (33%) had a single Class I DSA: modeling showed a positive AHG-CDC threshold of MFI > 10,000, with probability of a positive AHG-CDC of 0% (0/62) below and 73% (8/11) above this value (ROC AUC = 0.99). 109/220 samples (50%) contained multiple Class I DSA: modeling showed a positive threshold of MFI > 5000, with probability of a positive AHG-CDC of 3% (1/30) below and 72% (57/79) above this value (ROC AUC = 0.86). Applying logistic regression modeling further confirmed the distinct separation of thresholds for positive AHG-CDC in samples containing a single DSA versus multiple DSA with the limits defined above (p < 0.0001) (Figure 1).
Conclusion: For sera containing a single Class I DSA, the threshold for prediction of a positive AHG-CDC XM appears to be an MFI of 10,000, whereas this declines to an MFI of 5000 in sera with multiple anti-donor specificities. These values both differ markedly from the threshold for definition of DSA according to current consensus guidelines which serve as the basis for transplant recipient selection.
To cite this abstract in AMA style:
Lan J, Espino-Hernandez G, Gill J, Keown P. Probability of a Positive AHG-CDC Crossmatch Is Determined by the Number, Strength, and Specificity of Donor-Specific Antibodies [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/probability-of-a-positive-ahg-cdc-crossmatch-is-determined-by-the-number-strength-and-specificity-of-donor-specific-antibodies/. Accessed May 21, 2026.« Back to 2013 American Transplant Congress
