Primary Indications and Waitlist Status for Pediatric Living Donor Liver Transplants
K. Goli1, K. Hosek2, A. Rana3, J. Goss3, T. Miloh3
1Rice University, Houston, TX, 2Texas Children's Hospital, Houston, TX, 3Abdominal Transplant, Baylor College of Medicine/Texas Children's Hospital, Houston, TX
Meeting: 2019 American Transplant Congress
Abstract number: 457
Keywords: Living-related liver donors, Pediatric, Split-liver transplantation, Waiting lists
Session Information
Session Name: Concurrent Session: Liver: Living Donors and Partial Grafts
Session Type: Concurrent Session
Date: Tuesday, June 4, 2019
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:30pm-3:42pm
Presentation Time: 3:30pm-3:42pm
Location: Room 312
*Purpose: Since 1989, living donor liver transplantation (LD) has been used for a selected population and has shown improved long-term graft survival. The aim was to compare LD and deceased donor liver transplants (DD) in children from UNOS database by studying indications, status at listing, and graft/patient survival across regions.
*Methods: The UNOS database was queried for pediatric (age <18yrs) liver transplant (LT) recipients in years 2002-2018 (retransplants and multiorgan recipients excluded). Chi square and t test were used for univariate analysis. After adjusting for 55 covariates, graft survival probabilities were computed as days to graft failure with Cox proportional hazard function. Patient survival was estimated with Kaplan-Meier. Significance tests (α≤0.05) were two-tailed.
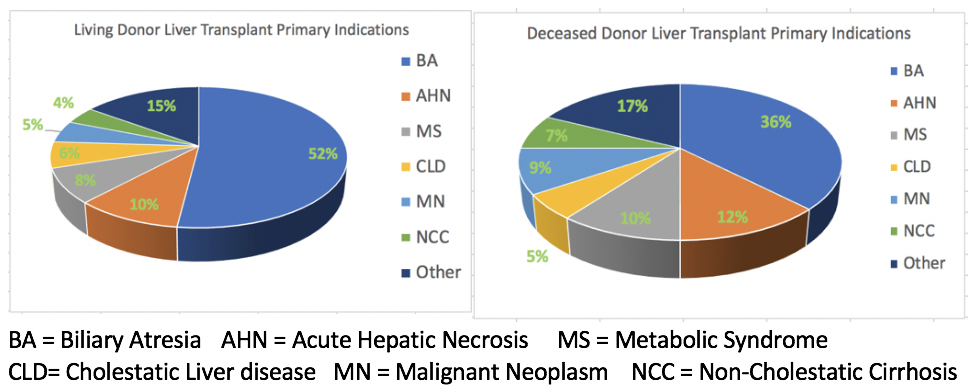
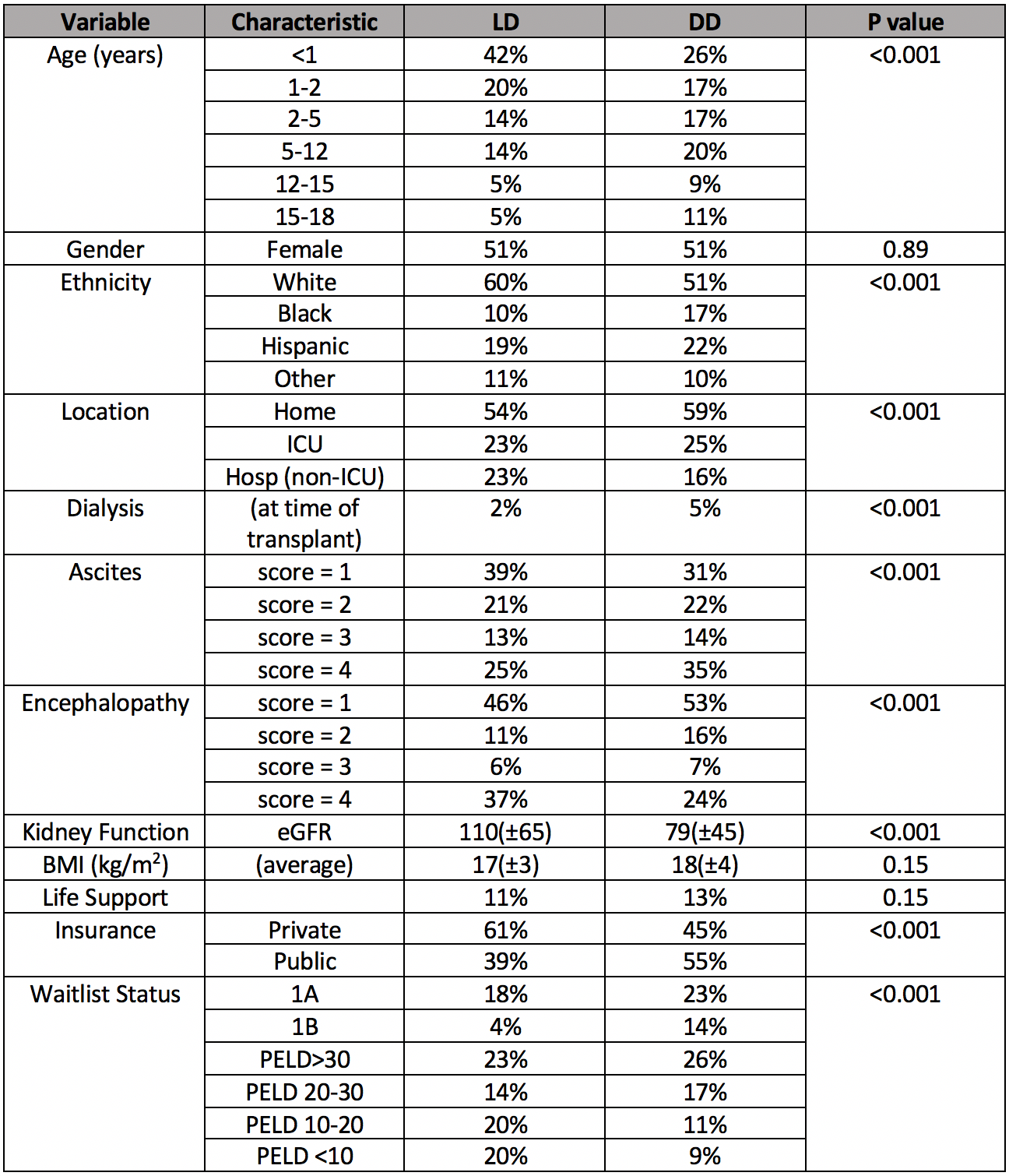
*Results: From 2002-2018, an average of 60 LDLTs were performed per year in children, with 998 LD out of 8236, representing 12% of total LT. Over half of the primary indications in LD were biliary atresia, compared with 36% of DD. Other indications are presented (Figure 1) (P<0.001). Demographics and waitlist status are presented (Table 1). All times during follow-up, LD experienced lower risk of graft loss than DD (P=0.0005). Adjusting for 55 covariates, LD had significantly lower risk of graft failure than DD in the following UNOS regions: 1 (HR:0.683(0.478-0.974)), 2 (HR:0.682(0.531-0.875)), 4 (HR:0.737(0.567-0.958)), 5 (HR:0.613(0.483-0.779)), 6 (HR:0.573(0.359-0.916)). For LD compared to DD, adjusted graft survival (HR:0.82 (P=0.04)) and unadjusted patient survival (P=0.0008) were significantly greater.
*Conclusions: In children, primary indications (i.e. biliary atresia) and other indicators (i.e. age<2yrs, White ethnicity, ascites score=1, private insurance, PELD<20) correlated with higher probability of receiving LD. LD graft survival differs across UNOS regions. Graft and patient survival are higher for LD. Clinical benefit may exist in exploring LD options in other groups (i.e. across ethnicities and insurance status).
Figure 1: Primary Indications for Pediatric LD and DD
Table 1: Demographics and Clinical Data at Time of LT
To cite this abstract in AMA style:
Goli K, Hosek K, Rana A, Goss J, Miloh T. Primary Indications and Waitlist Status for Pediatric Living Donor Liver Transplants [abstract]. Am J Transplant. 2019; 19 (suppl 3). https://atcmeetingabstracts.com/abstract/primary-indications-and-waitlist-status-for-pediatric-living-donor-liver-transplants/. Accessed May 26, 2026.« Back to 2019 American Transplant Congress