Primary Graft Dysfunction Post Heart Transplantation: Is There Greater Risk for De Novo Donor-Specific Antibody Development?
Cedars-Sinai Heart Institute, Los Angeles
Meeting: 2017 American Transplant Congress
Abstract number: C101
Keywords: Antibodies, Heart transplant patients
Session Information
Session Name: Poster Session C: Hearts and VADS: All Topics
Session Type: Poster Session
Date: Monday, May 1, 2017
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall D1
Background: Primary graft dysfunction (PGD) may occur in up to 30% of heart transplant recipients and has been associated with increased mortality. The development of de novo donor specific antibodies (DSA) has also been associated with increased mortality and risk of rejection after heart transplantation. It has not been established whether patients with PGD are predisposed to development of de novo DSA post-heart transplant.
Methods: Between 2011 and 2015 we assessed 494 patients for development of PGD at our single center. 43 patients were identified with PGD (left or bi-ventricular) and were compared to a non-PGD control. Outcomes for each group included 1-year survival, 1-year freedom from non-fatal major adverse cardiac events (NF-MACE: defined as myocardial infarction, congestive heart failure, percutaneous cardiac intervention, placement of pacemaker/defibrillator, stroke), 1-year freedom from treated rejection, and 1-year freedom from de novo DSA development. The time to DSA development was also assessed.
Results: Patients with PGD had longer ischemic times compared to control (181.1 ± 58.3 vs 154.8 ± 57.2 min, p= 0.005). PGD was associated with reduced 1-year survival, 1-year freedom from de novo DSA, antibody-mediated rejection, and NF-MACE compared to the non-PGD control (Table). These patients were more sensitized prior to heart transplantation and had a greater prevalence of anti-thymocyte globulin induction therapy post-heart transplant. There was no significant difference in the average time to de novo DSA development in the PGD and non-PGD cohorts (4.4mo± 5.2 vs 4.6mo± 4.2, p=0.806). Both groups appear to have a greater occurrence of Class II de novo DSA upon initial development (71.4% vs 76.9%, p=1.0).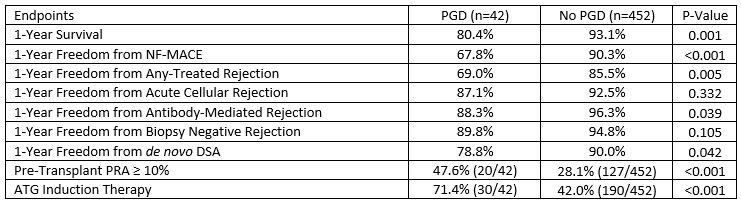 Conclusion: Patients with PGD post-heart transplant appear to be at greater risk for de novo DSA development and antibody-mediated rejection. Further studies are warranted with a larger population size and longer follow-up to confirm these results.
Conclusion: Patients with PGD post-heart transplant appear to be at greater risk for de novo DSA development and antibody-mediated rejection. Further studies are warranted with a larger population size and longer follow-up to confirm these results.
CITATION INFORMATION: Patel J, Kittleson M, Czer L, Aintablian T, Kao T, Geft D, Chang D, Esmailian F, Kobashigawa J. Primary Graft Dysfunction Post Heart Transplantation: Is There Greater Risk for De Novo Donor-Specific Antibody Development? Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Patel J, Kittleson M, Czer L, Aintablian T, Kao T, Geft D, Chang D, Esmailian F, Kobashigawa J. Primary Graft Dysfunction Post Heart Transplantation: Is There Greater Risk for De Novo Donor-Specific Antibody Development? [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/primary-graft-dysfunction-post-heart-transplantation-is-there-greater-risk-for-de-novo-donor-specific-antibody-development/. Accessed May 16, 2026.« Back to 2017 American Transplant Congress
