Pre-Transplant Assessment of AMR Risk by Novel Donor/recipient Non-HLA Variants
1UCSF, San Francisco
2Johns Hopkins University, Baltimore.
Meeting: 2018 American Transplant Congress
Abstract number: 25
Keywords: Kidney transplantation, Rejection
Session Information
Session Name: Concurrent Session: Kidney Acute Antibody Mediated Rejection
Session Type: Concurrent Session
Date: Sunday, June 3, 2018
Session Time: 2:30pm-4:00pm
 Presentation Time: 3:42pm-3:54pm
Presentation Time: 3:42pm-3:54pm
Location: Room Hall 4B
Transplant (tx) injury from antibody mediated (AMR) and T cell mediated (CMR) rejection, limits indefinite survival after HLA mismatched organ tx. AMR is incompletely diagnosed as donor/recipient (D/R) matching is only limited to the HLA locus and critical nHLA immunogenic antigens remain to be identified. We have developed an integrative computational approach leveraging D/R exome sequencing and gene expression to predict risk of post-tx AMR and CMR, by pre-tx assessment of a single blood sample from the D/R pair. Rigorous statistical analysis on 28 highly annotated D/R kidney tx pairs before tx, where the recipients developed biopsy confirmed rejection after tx, identified a significantly higher number of mismatched nHLA variants in AMR (p = 0.02), 123 variants associated with post-tx AMR risk (p < 0.001; Fischer's exact test), from which a subset of 65 variants identified recipients likely to develop AMR with very low error rate (random forest).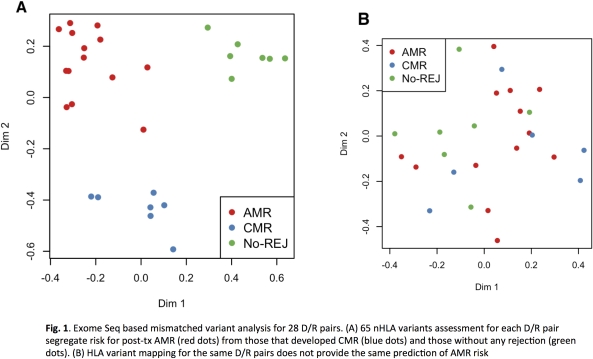 These variants are functionally highly relevant to the rejection process as they relate to genes and/or eQTLs that are enriched in genes expressed in kidney and vascular endothelium and underlie the immunobiology of graft rejection. In addition to current D/R HLA mismatch evaluation, additional D/R mismatch nHLA analysis for the 65 variants, can enhance pre-tx D/R pair risk stratification for post-tx recipient AMR risk. This provides an AMR risk-score for each D/R pair even before engraftment of the tx organ, allowing for selective optimization for the D/R pair with lowest AMR and CMR risk, when multiple D/R pairs are available in the context of living donation. In the case of deceased donation, the AMR risk score for D/R pair can drive immunosuppression selection. This innovative study design is applicable in all solid organ tx, where the impact of mitigating AMR on graft survival may be greater, with considerable benefits on improving human morbidity and mortality and opens the door to precision immunosuppression delivery and extended tx survival.
These variants are functionally highly relevant to the rejection process as they relate to genes and/or eQTLs that are enriched in genes expressed in kidney and vascular endothelium and underlie the immunobiology of graft rejection. In addition to current D/R HLA mismatch evaluation, additional D/R mismatch nHLA analysis for the 65 variants, can enhance pre-tx D/R pair risk stratification for post-tx recipient AMR risk. This provides an AMR risk-score for each D/R pair even before engraftment of the tx organ, allowing for selective optimization for the D/R pair with lowest AMR and CMR risk, when multiple D/R pairs are available in the context of living donation. In the case of deceased donation, the AMR risk score for D/R pair can drive immunosuppression selection. This innovative study design is applicable in all solid organ tx, where the impact of mitigating AMR on graft survival may be greater, with considerable benefits on improving human morbidity and mortality and opens the door to precision immunosuppression delivery and extended tx survival.
CITATION INFORMATION: Pineda S., Sigdel T., Chen J., Jackson A., Sirota M., Sarwal M. Pre-Transplant Assessment of AMR Risk by Novel Donor/recipient Non-HLA Variants Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Pineda S, Sigdel T, Chen J, Jackson A, Sirota M, Sarwal M. Pre-Transplant Assessment of AMR Risk by Novel Donor/recipient Non-HLA Variants [abstract]. https://atcmeetingabstracts.com/abstract/pre-transplant-assessment-of-amr-risk-by-novel-donor-recipient-non-hla-variants/. Accessed May 15, 2026.« Back to 2018 American Transplant Congress
